By focusing on the five key areas we’ve identified below you can ensure the AI you are evaluating to integrate into your quality and manufacturing processes meets regulatory requirements. At MasterControl, we understand the effort and time/resource investment required to thoroughly evaluate AI for regulatory compliance. That’s why we’ve done the work for you with the AI tools we build and...
Topics should cover the financial impact of data management systems on drug development, manufacturing, and distribution; the basic computer system life cycle model as well as the activities and software quality assurance practices in each phase; and the controls and methods necessary to maintain data integrity and security.
The FDA recommended CSA seeks to speed Computer System Validation (CSV) without sacrificing quality, but there’s a lot to know before you get started. This Kneat eBook, authored by CSV expert Darren Geaney, will give you a foundation of understanding so you can take the first step in your CSA journey with confidence.
By focusing on the five key areas we’ve identified below you can ensure the AI you are evaluating to integrate into your quality and manufacturing processes meets regulatory requirements. At MasterControl, we understand the effort and time/resource investment required to thoroughly evaluate AI for regulatory compliance. That’s why we’ve done the work for you with the AI tools we build and...
The FDA recommended CSA seeks to speed Computer System Validation (CSV) without sacrificing quality, but there’s a lot to know before you get started. This Kneat eBook, authored by CSV expert Darren Geaney, will give you a foundation of understanding so you can take the first step in your CSA journey with confidence.
The increasing digitalization of the pharmaceutical and medical device industry has created novel cybersecurity challenges, particularly with the rapid advancement of artificial intelligence (AI) technologies. This article examines the dual nature of AI as both a potential threat vector and a powerful defensive tool.
Pharma 4.0™ is a reference framework tailored to the pharmaceutical industry, guiding its digital transformation. Although many of today’s processes generate sufficient data to enable advanced use cases, structured guidance for transformation is often lacking. To address this, the ISPE Pharma 4.0™ Subcommittee on Process Data Maps and Critical Thinking has introduced an approach to help...
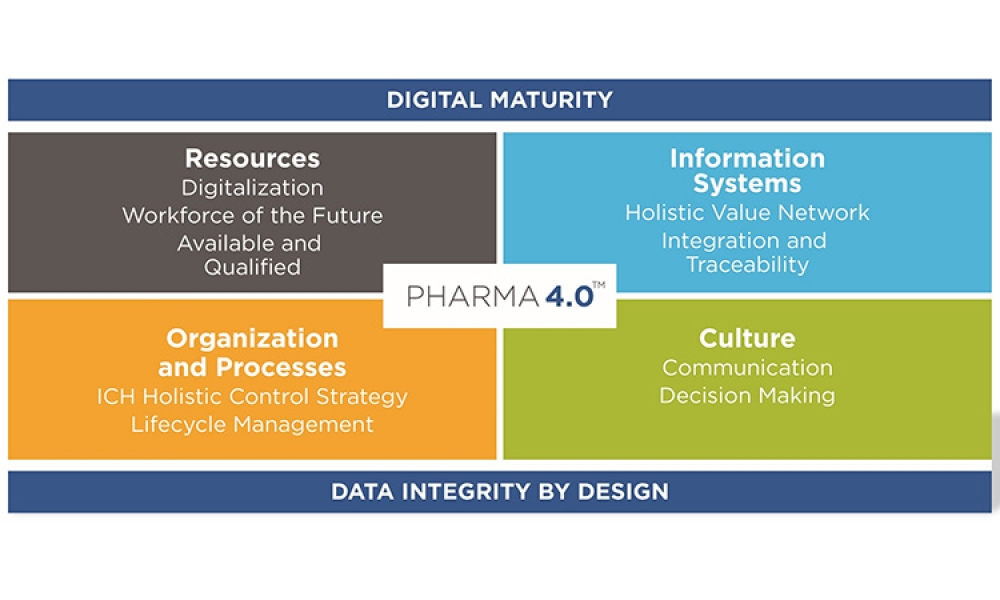
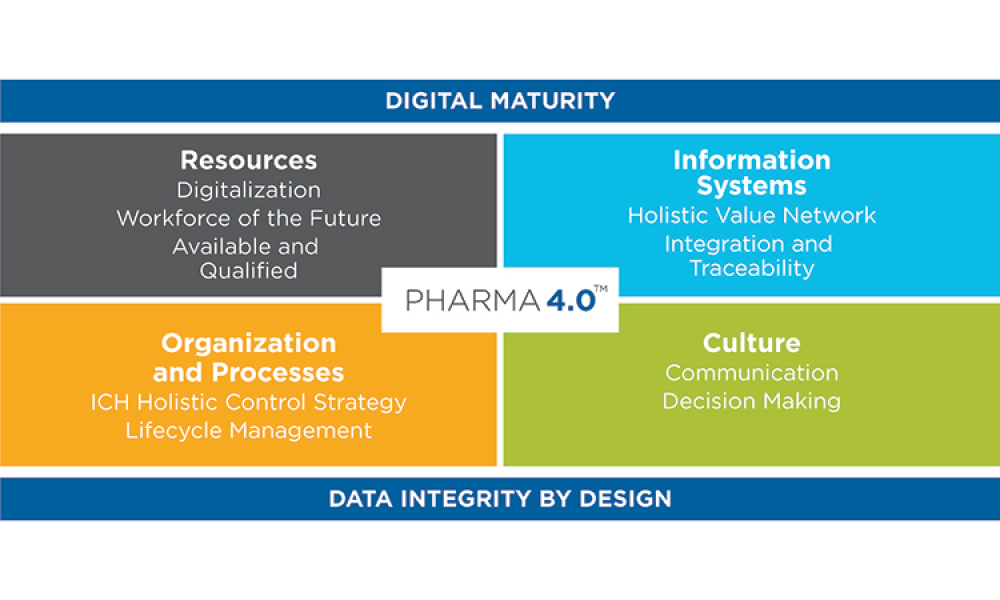
The ISPE Baseline® Guide Vol 8: Pharma 4.0™ was published in December 2023 to help accelerate the adoption of digital transformation. In quoting the opening narration of the original 1960s Star Trek television series, Christian Wölbeling, Founder and Chair of the ISPE Pharma 4.0™ Community of Practice, likes to say, “To boldly go where no one has gone before.”
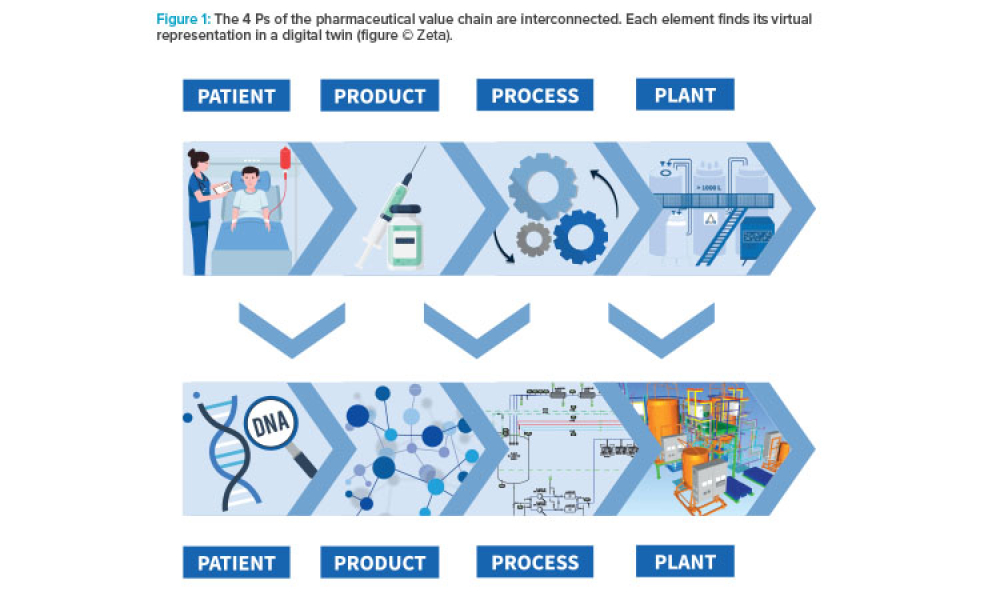
With the help of Digital Twins, companies can achieve greater certainty and precision in making informed decisions at various stages of the product lifecycle, driven by a deep understanding of underlying Critical Business Parameters (CBPs).
Informed consent aims to provide clinical trial participants with transparent education about the trial’s objectives, potential risks and benefits, and procedural requirements. As clinical trial designs evolve to include decentralized and adaptive elements, the informed consent process becomes increasingly complex. In response, this article explores the potential application of AI-powered...
Pharmaceutical companies can no longer rely on traditional drug development methods to keep pace with complex therapies and evolving regulatory demands. This Industry Insight examines how digital transformation, combined with Quality by Design (QbD) principles, improves efficiency, ensures compliance, and enhances product quality from the start.
Pharma 5.0™ is here, and AI is transforming the life sciences industry. Are your QA and validation processes ready? Kneat’s eBook, authored by AI expert Daniel Tobin, offers a comprehensive guide to leveraging AI for compliance, efficiency, and innovation at your organization.
In the fast-evolving life sciences sector, lab design, functionality, and technology integration are key to efficiency, collaboration, and success. With rising demand, companies are embracing agile, science-first strategies. A major shift is the integration of AI and machine learning, transforming research and pharmaceutical labs. These advancements streamline operations and drive innovation,...
The landscape of clinical trials has been transformed in a post-pandemic world. In July, ISPE released the second edition of ISPE GAMP® Good Practice Guide: Validation and Compliance of Computerized GCP Systems and Data – Good eClinical Practice. It addresses managing complexities associated with decentralized clinical trials, the benefits and challenges of using open-source software, and...
This article describes a practical and pragmatic approach to the management of computerized system life cycle and information technology (IT) process records. The objective is to effectively achieve and maintain compliant GxP-regulated systems that are fit for intended use, and to support patient safety, product quality, and data integrity.
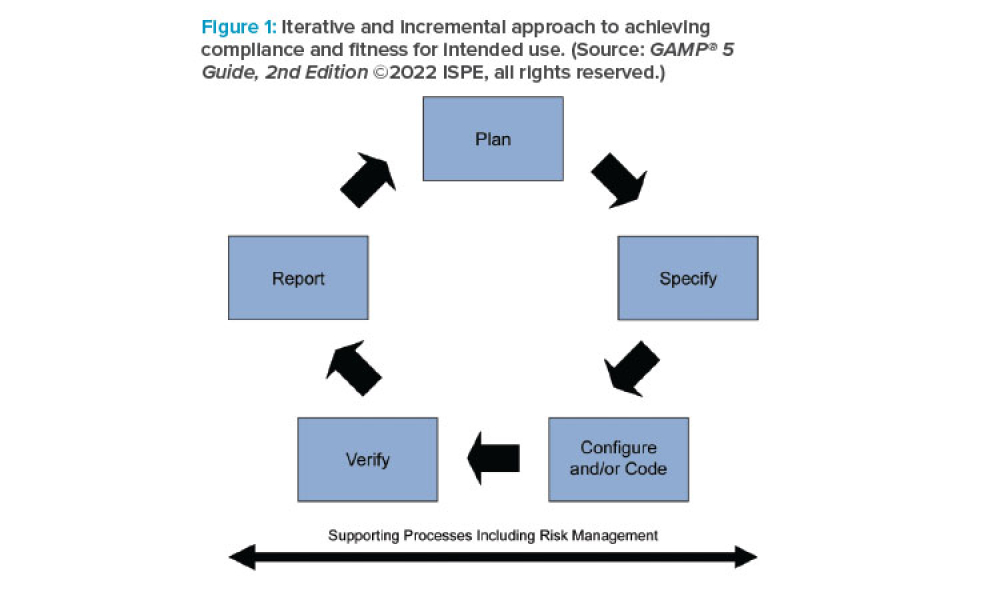
Risk assessment is essential, however, risk assessment is of limited value if it is not conducted by a team with the necessary process, product, and functional understanding. Conducting risk assessments prematurely may lead to invalid assessment of overall risks. Conducting risk assessments too late will limit the opportunity to address design flaws and effectively test processes and...
This special anniversary article addresses the history and milestones that define the GAMP Community of Practice (CoP). In celebration of the 25th anniversary of the creation of GAMP Americas, we reflect on the vital role GAMP Americas has played in that journey. We commemorate key accomplishments of its members, share recent activities, and look ahead to the future of GAMP Americas.
Technology is advancing at an extraordinary rate. Industries are benefiting from automation and AI. As technological developments continue to reform the way industries run, the integration of AI and machine learning technologies in the life sciences industry is redefining the traditional approach to commissioning, qualification, and validation (CQV) in pharmaceutical manufacturing.
The integration of artificial intelligence (AI) and machine learning (ML) into bioprocess development represents a rapid shift in the way discovery, development, optimization, and production of biological products are approached.
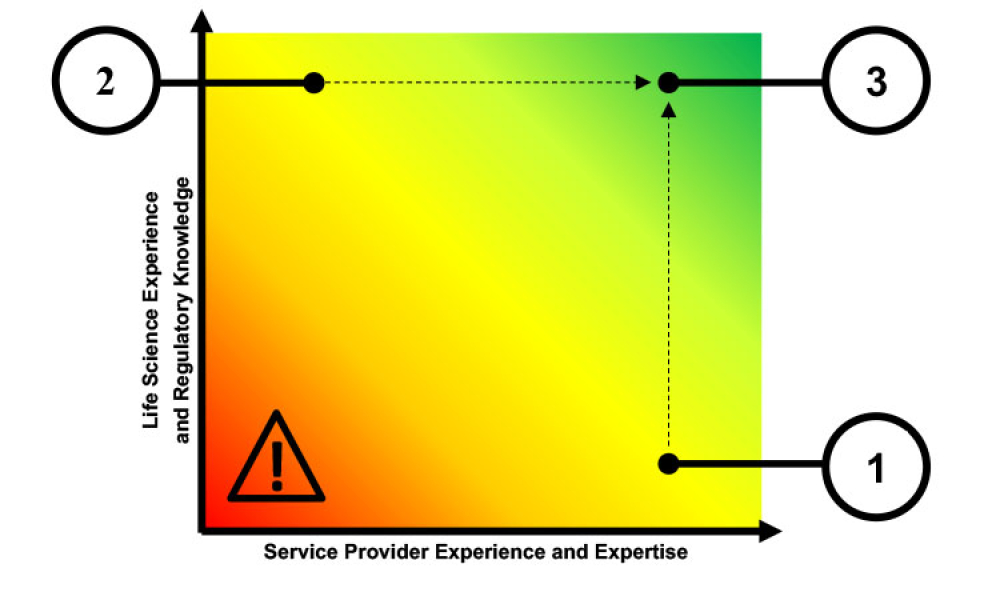
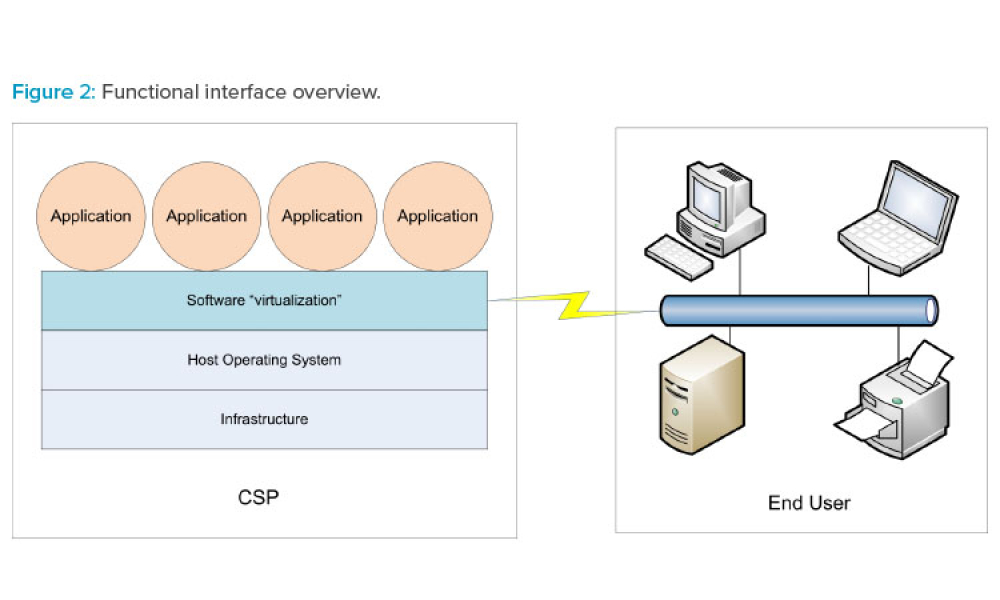
Traditionally, a regulated company is accountable for all aspects of their infrastructure qualification and application validation. With the introduction of public cloud service providers (CSPs), part of that technical responsibility has shifted to a cloud supplier, making supplier assessment and supplier management more important than ever—even though the regulated company is still...
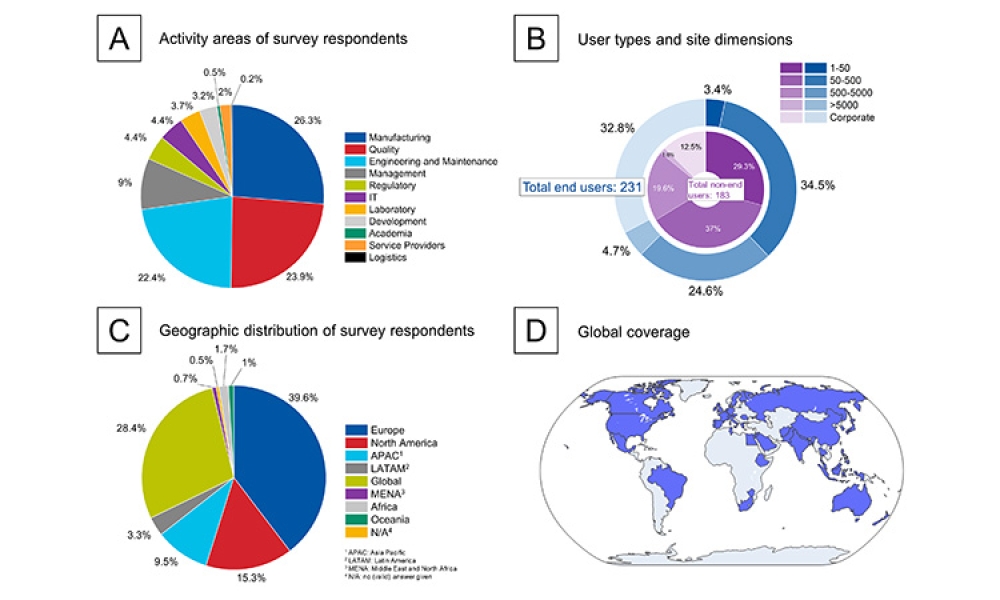
This article summarizes the key findings from the 7th Pharma 4.0™ Survey, conducted in 2023. It explores the demographics of the survey respondents, maturity levels of Pharma 4.0™ adoption, enabling technologies being leveraged, anticipated benefits, and challenges encountered.
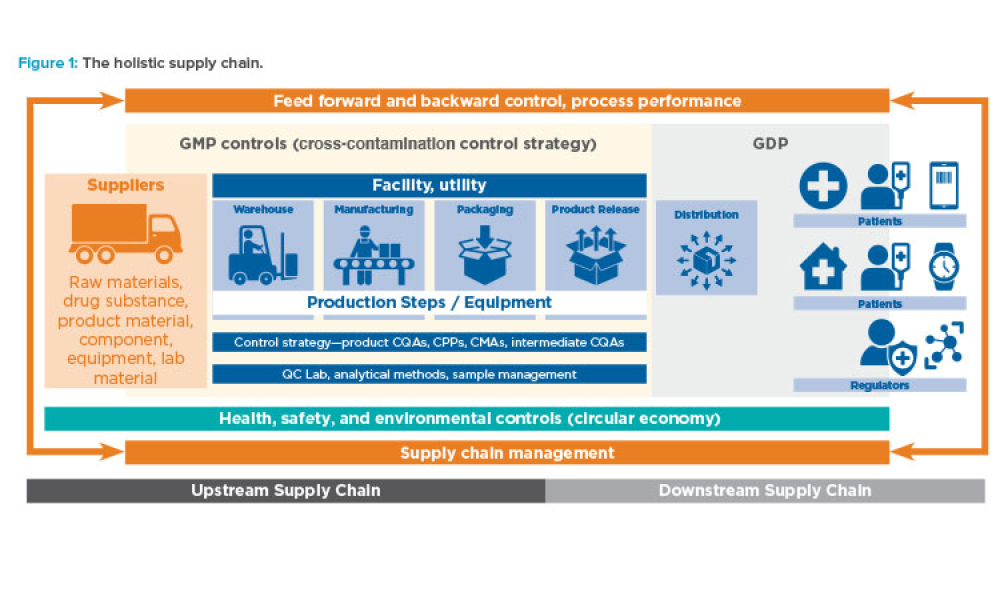
The pharmaceutical industry faces considerable challenges throughout the development, manufacturing, and supply of medicines, largely due to the intricate and divergent global regulatory landscape. The adoption of structured data standards and utilization of cloud-based platforms offer immense potential to overcome these challenges by facilitating faster and more efficient global...
Computer software assurance (CSA) has been discussed widely in industry over the past five years. While the principles are well understood and welcomed, until now some of the practical detail on how exactly to implement CSA into an organization has been missing.
The pharmaceutical industry stands at the precipice of a revolution as emerging digital technologies provide new opportunities to boost productivity through continuous process improvements. The Pharma 4.0™ framework, an adaptation of the broader Industry 4.0 movement, aims to transform how drugs are produced and delivered.
Implementing advanced automation technologies is a strategic move that can amplify the positive outcomes of environmental, social, and governance (ESG) initiatives. By leveraging ESG initiatives, pharmaceutical companies can enhance their competitive edge and contribute positively to global sustainability efforts.
Artificial intelligence (AI) is used by pharmaceutical and biotech companies, providing support from drug discovery through manufacturing. The nature of AI and concerns of bias, privacy, transparency, and security in a regulated industry necessitate a governance framework to ensure concerns are controlled using “guardrails.” These guardrails ensure the quality, privacy, and security of data...
One of the primary technical objectives of the ISPE Pharma 4.0™ Plug and Produce Working Group is to assist the Pharma 4.0™ digital transformation by enabling seamless integration and interoperability between all systems components and operational technology to advance the overall digital maturity toward predictive and adaptable operations.
Digital display labels (DDLs) offer an alternative solution to eliminate manual relabeling in the clinical supply chain, optimizing label content updates through a simple, system-controlled approach while providing new, uncharted opportunities. With increased efficiency in making regulatory-compliant changes and enhanced flexibility in the clinical supply chain, DDL technology has the...
In 2022, the US Food and Drug Administration (FDA) issued their draft guidance “Computer Software Assurance for Production and Quality System Software”
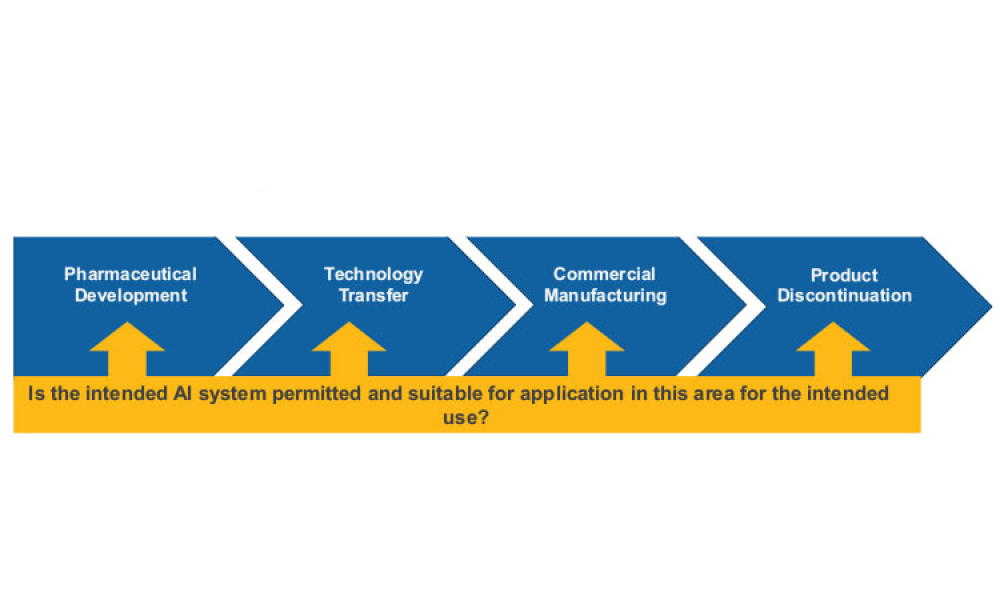
Stakeholders across industries are becoming accustomed to using information technology (IT) systems, applications, and business solutions that feature artificial intelligence (AI) and machine learning (ML). Even though some of these uses show phenomenal performance, thorough risk management is required to ensure quality and regulatory compliance are met within the life sciences industry. By...
Due to the growing digitalization of the industry, we are highly dependent on information technology (IT) systems and data. The basic ability to execute our pharmaceutical business and decision-making processes relies on the permanent availability of these IT systems and data to ensure compliance and efficiency of our business operations. But numerous factors—including criminal activities,...
IT infrastructure has traditionally been provisioned using a combination of scripts and manual processes. This manual approach was slow and introduced the risk of human error, resulting in inconsistency between environments or even leaving the infrastructure in an unqualified state. In this article, we investigate some fundamental advantages of using Infrastructure as Code (IaC) for...
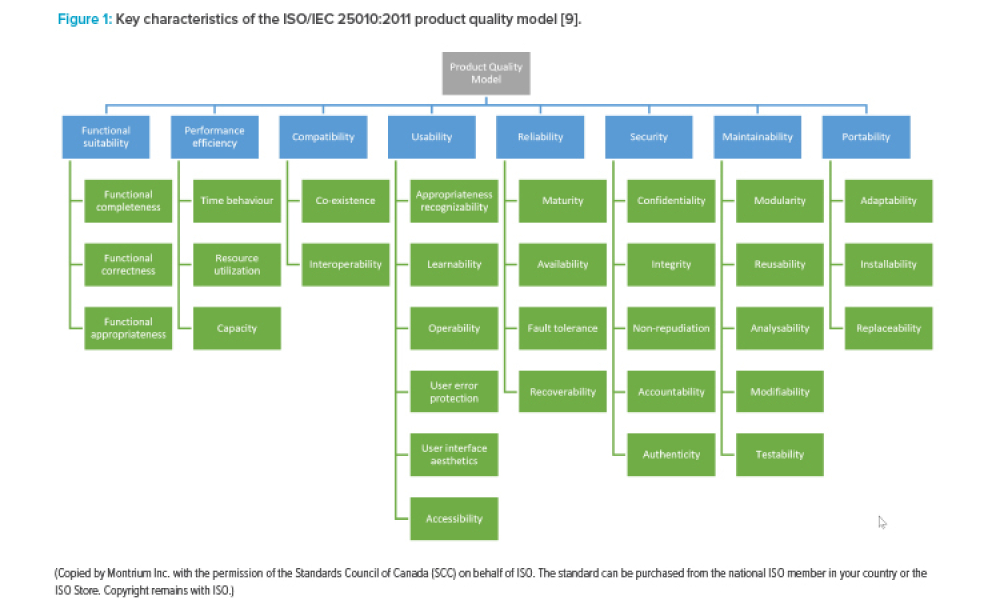
This article provides a brief introduction into the standards and regulations for medical devices. It compares the ISPE GAMP® 5 Guide: A Risk-Based Approach to Compliant GxP Computerized Systems (Second Edition) and applicable ISPE GAMP Good Practice Guides against the relevant regulations and standards for the development of software for medical devices and demonstrates GAMP® 5 Second...
Innovation is an integral part of corporate strategy. Initiatives to facilitate innovation are continually developed and pursued. The 2022 ISPE Pharma 4.0™ Emerging Leader (EL) Hackathon was designed based on innovator needs and provided a hands-on blueprint manufacturing exercise. Over a period of two days and facilitated by 40 subject matter experts, coaches, and jury members, 50...
The fifth Pharma 4.0™ conference was held December 2022 in Vienna, Austria, in combination with the Aseptic Processing conference. Nearly 500 participants attended either in person or online to learn about the latest developments.
ChatGPT and other large language models are positioned to change the world. They can also shift acceptance and prevalence of machine learning solutions in regulated industries in general. However, their arrival requires reconsiderations on risks, quality assurance, and validation from a GxP perspective.
Funded by the European Commission from 2019, the Smart Pharmaceutical Manufacturing Project (SPuMoNI)
In the context of data integrity, data flows are essential. The FDA, PIC/S, and WHO have all emphasized the importance and benefits of data flows in their guidance on data integrity. The key to data integrity compliance is a well-functioning data governance system
We will show how continuous, real-time capturing of data with immediate data analysis by an ML algorithm can improve control over a critical quality attribute. The ML-analyzed data provides the evidence for validation of the change by demonstrating more control over the process along with a decrease in process risks.
Recent advances in artificial intelligence (AI) have led to its widespread industrial adoption, with machine learning (ML) algorithms demonstrating advances in performance in a wide range of tasks. However, this comes with an ever-increasing complexity of the algorithms used, rendering such systems more difficult to explain.
ISPE’s GAMP® 5: A Risk-Based Approach to Compliant GxP Computerized Systems (Second Edition) (GAMP® 5 Guide, 2nd Edition) maintains the principles and framework of the first edition and updates their application in the modern world, including the increased importance of service providers, evolving approaches to software development, and expanded use of software tools and...
In this article, potential Pharma 4.0™ technological solutions that can enhance continuous process verification (CPV) 4.0 are discussed. The necessary paradigm shift will allow companies to predict deviations more accurately, perform root cause...
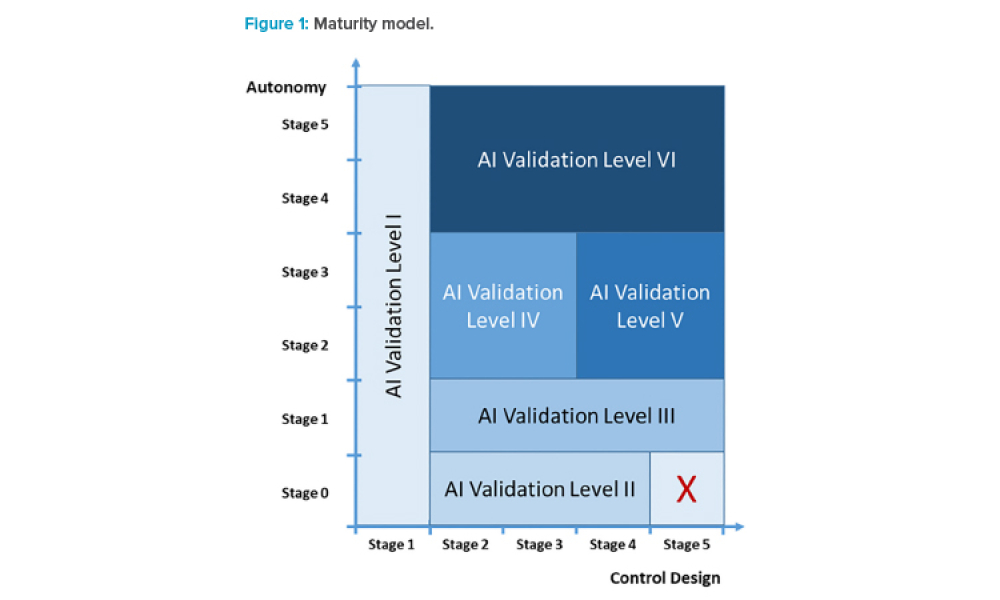
This article explores life-cycle activities for machine learning (ML) within regulated life sciences. It positions and contextualizes the life cycle and management of the machine learning subsystem or components within a wider system life cycle. It also gives general descriptions and guidance illustrated by a case study demonstrating a machine learning application to medical image recognition,...
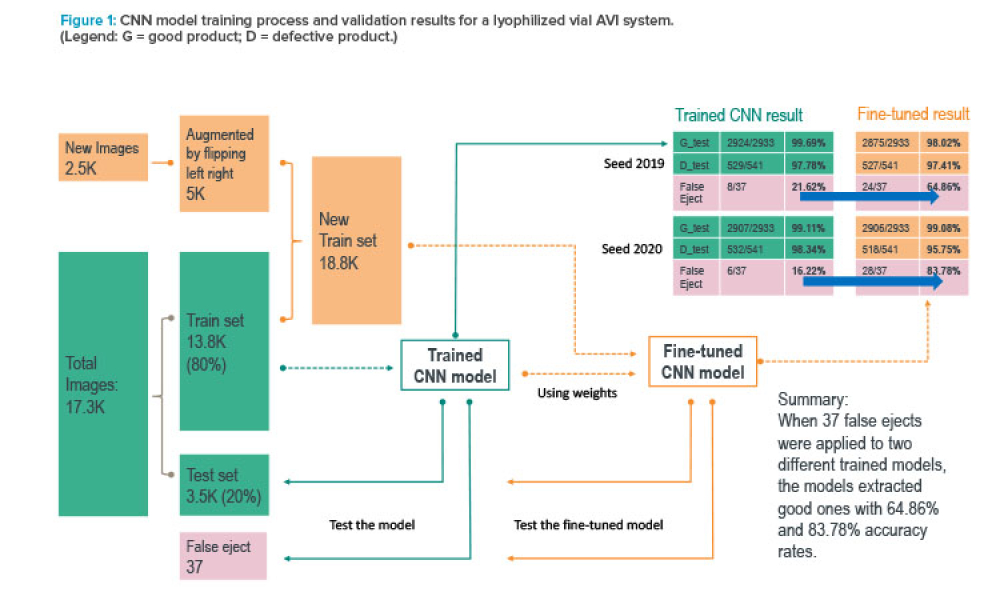
This artificial intelligence (AI) retrofit project was a unique approach to implementing AI technology in a pharmaceutical environment within three months. This project tackles a commonly known industry challenge by integrating AI into an existing automatic visual inspection (AVI) machine. The proof of value allowed us to benchmark added value through AI compared with state-of-the-art...
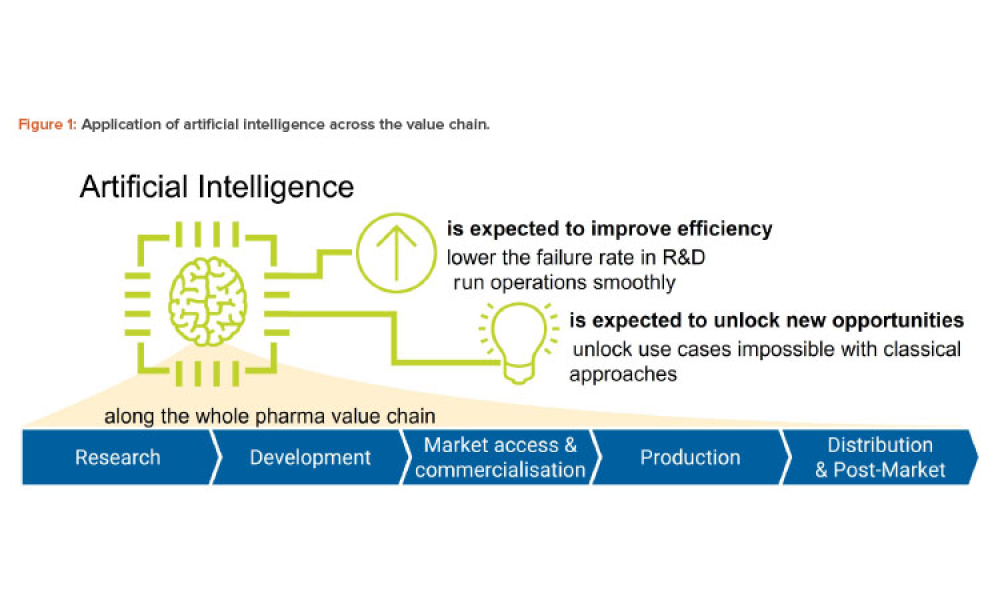
Artificial intelligence (AI) has the potential to benefit the pharmaceutical industry and its GxP-regulated areas. Several pharmaceutical companies are currently running digital pilots; 90% of large pharmaceutical companies have initiated AI projects.
The life cycle approach to process validation stresses the need for continued monitoring of process performance to ensure that the manufacturing process remains stable and predictable, i.e., in a state of control. This life cycle stage is known as continued process verification (CPV) or ongoing process verification (OPV).
As adoption of cloud technology continues to increase across the life sciences industry, so too does the need to establish a standardized and pragmatic approach for ensuring the quality of software applications used in support of GxP data and associated processes. This article focuses on the application level and the growing use of software as a service (SaaS) within the life sciences...
Artificial intelligence (AI) has become one of the supporting pillars for digitalization in many areas of the business world. The pharmaceutical industry and its GxP-regulated areas also want to use AI in a beneficial way. Several pharmaceutical companies are currently running digital pilots, but only a small fraction follows a systematic approach for the
This article aims to refresh information on open-source software (OSS) within regulated computerized systems that was first discussed in an article in May-June 2010 Pharmaceutical Engineering®. The adoption of OSS advanced since then, and the article explores the importance of recognizing when an organization is relying on OSS and the benefits and risks this brings from a GAMP® 5...
Developing comprehensive digital solutions is crucial for the entire value creation process for pharmaceuticals. A holistic view of the interrelations of product, production process, and plant is becoming increasingly significant. In this context, the application of model-based technologies provides support in drug development, process scale-up, and manufacturing. Furthermore, it accelerates...
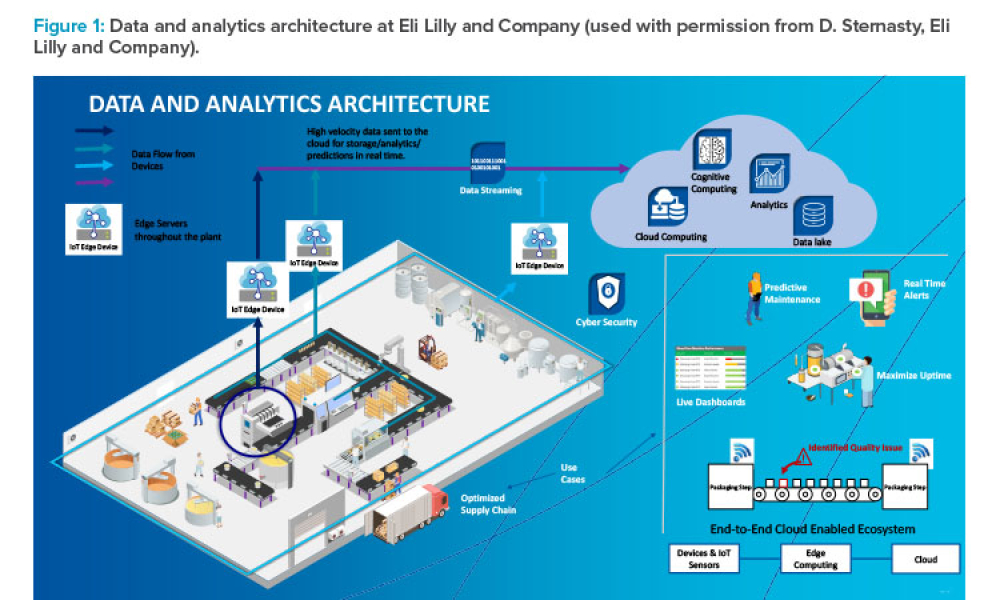
Industry 4.0 applications in biopharma involve the complete spectrum of data science throughout the entire product life cycle of many disparate entity types. Tools such as digitalization, modern data science, and the industrial internet of things (IIoT) exist now, and examples from other industries such as Siri and Alexa, face identification, and self-driving cars can guide their...
Use of software as a service (SaaS) applications within the life sciences industry is growing. This article reviews the issues and challenges faced by SaaS providers and identifies the qualities of successful providers for this highly regulated industry.
This second of a two-part series explores digital transformation and digitalization in the biopharmaceutical industry with information about how data science enables digitalization along the product life cycle. (Part 1 was published in the March-April 2021 issue of Pharmaceutical Engineering.
This article provides a beginner’s overview of how organizations can achieve a state of preparedness (readiness) for inspections, with a specific focus on IT systems.
Cloud computing can be described as networked access and utilization of configurable computing resources such as data and information storage, processing capabilities, applications, and other services on computerized systems provided and/or maintained by a remote organization. As life sciences companies consider the advantages and costs of utilizing cloud services, they first need to invest...
Real-world evidence (RWE) is clinical evidence regarding the usage and potential benefits or risks of a medical product derived from analysis of real-world data (RWD) relating to patient health status and the healthcare delivery.
This article focuses on pragmatic quality- and risk-based approaches to IT infrastructure. It covers recommendations made by a US FDA/industry team linked to the US FDA Center for Devices and Radiological Health (CDRH) Case for Quality initiative
Industry 4.0 is the recent movement toward intelligent automation technology. In this new era, the integration of modern manufacturing skills and novel information technologies plays an important role on economic competitiveness.
The ISPE Pharma 4.0™ Special Interest Group (SIG) launched in 2015 to provide a road map for new challenges of digitalization, Industry 4.0, and the smart factory. The Special Interest Group addresses how pharmaceutical industry stakeholders, including regulatory authorities, can achieve benefits from
The fourth Industrial Revolution (also known as Industry 4.0) is the era of smart machines, storage systems, and production plants that can autonomously exchange information, trigger actions, and control operations free of any human intervention. To ensure future success in the delivery of therapeutic medicines to patients, it is imperative that the pharmaceutical industry move deeper into the...
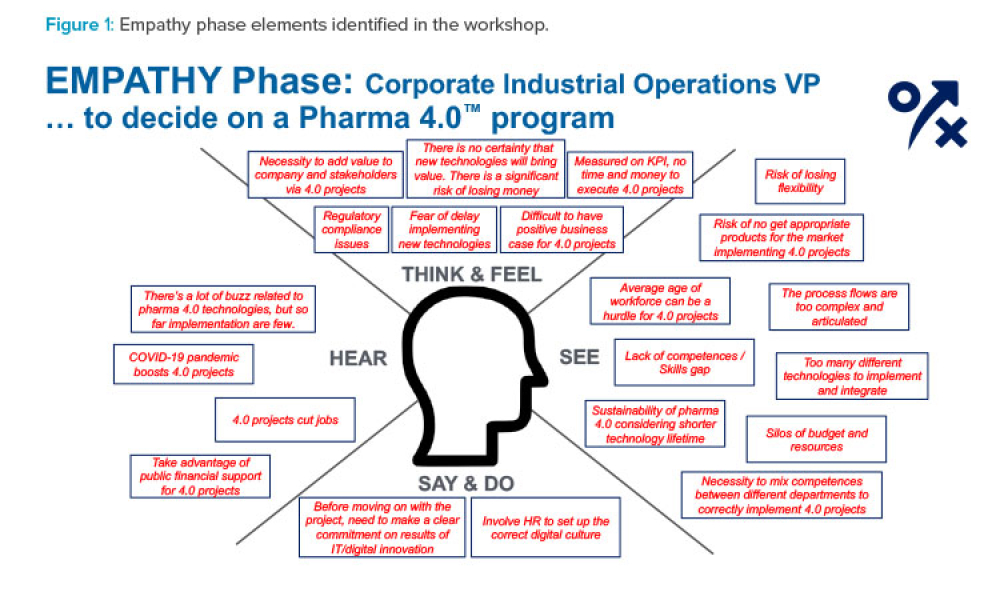
During the ISPE Pharma 4.0™ Virtual Conference, the Management Communication working group of the ISPE Pharma 4.0™ Special Interest Group (SIG) held a workshop to support ISPE members in pitching,...
Through this difficult time of the COVID-19 pandemic, ISPE has remained active. At the 2020 ISPE Pharma 4.0™ Virtual Conference, 17–18 November, 174 attendees gathered online to discuss and learn about...
Understanding and utilization of Pharma 4.0™ technologies will be critical for students and Emerging Leaders (ELs) as they develop in their careers in the pharmaceutical and life sciences industries. To learn more, I spoke with Edoardo Schiraldi, an Emerging Leader based in Florence, Italy, who works as a Corporate R&D Business Solutions Specialist with Menarini Group, about the “pillars”...
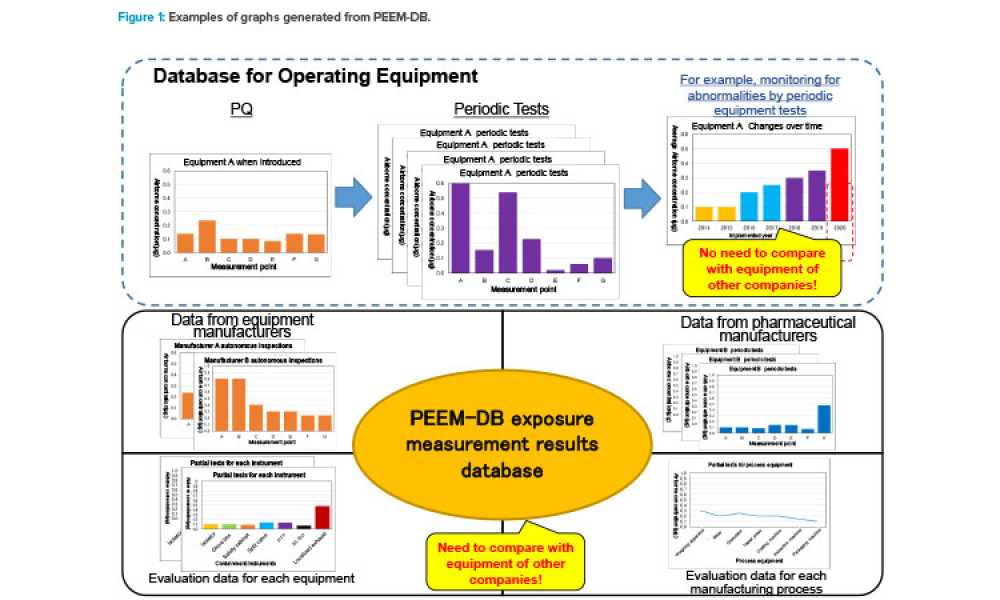
This article describes the Pharmaceutical Equipment Exposure Measurement Database (PEEM-DB), which was launched in July 2019 by the ISPE Japan Affiliate for its members. PEEM-DB is offered as a tool for rationally advancing optimal containment equipment settings by collecting exposure measurement results for...
Pharmaceutical companies rely on automated vision inspection (AVI) systems to help ensure product safety. Although these systems overcome challenges associated with manual inspection, they can be hindered by limitations in their programming—if the system is programmed to consider every variation in inspection conditions, it is likely to falsely identify defects in safe products. This article...
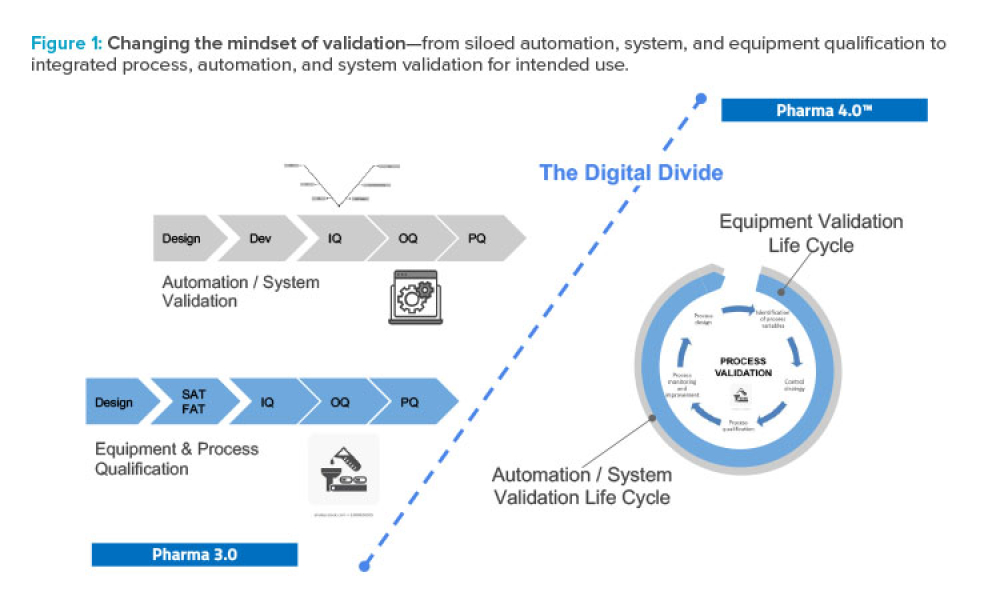
As Pharma 4.0™ increasingly becomes reality, our validation practices must change. We can no longer apply 20th-century thinking to 21st-century technology and resources. Validation must adapt to industry shifts from iterative to...
In the pharmaceutical industry, digitalization involves developing and implementing digital technologies at all levels of pharmaceutical operations. The aim is to transform the industry by capturing, analyzing, and using vast amounts of data collected from a wide range of sources to support research and development (R&D), clinical development, drug manufacturing, supply chain management,...
Between 2009 and 2019, the number of adverse events (AEs) for drugs and therapeutic biologic products recorded by the US FDA Adverse Event Reporting System (FAERS) increased more than 300%, from 490,032 to 2.19 million cases (as of 31 December 2019).
This article introduces the concept of robotic process automation (RPA) and discusses how the technology may be used within a GAMP® framework to support both non-GxP and GxP processes.
The Advanced Digital Design of Pharmaceutical Therapeutics (ADDoPT) project