About Pharmaceutical Engineering®

Editorial Mission & Focus
Pharmaceutical Engineering® is a technical magazine published by ISPE for its Members, who are engaged in the research, development, and manufacture of medicines and medical devices. Pharmaceutical Engineering® magazine covers all sectors of the global pharmaceutical industry, focusing on scientific and technical developments, regulatory initiatives, and innovative solutions for real-life challenges. All technical content is peer reviewed.
Practical articles and case studies highlight global best practices in engineering and design, product development, technology transfer, process development and scale-up, commercial manufacturing, quality and compliance, and product lifecycle management. Features and member editorials focus on new technology, quality management practices, and innovation. Other topics include biopharma, breakthrough therapies, next-generation manufacturing, quality metrics, quality risk management, supply chain integrity, and sustainability.
Technical articles
All technical article submissions are peer reviewed by an expert Member editorial board. Technical articles are published in the following categories:
Facilities and Equipment: Articles should discuss process equipment, the design of facilities, and utility systems that support the critical physical and chemical requirements of drug products, in addition to other aspects of the product specification. Topics could include design and construction/installation, commissioning and qualification, operation and maintenance, and controls and automation.
Information Systems: Topics should cover the financial impact of data management systems on drug development, manufacturing, and distribution; the basic computer system life cycle model as well as the activities and software quality assurance practices in each phase; and the controls and methods necessary to maintain data integrity and security.
Product Development: Articles should explore the interactions of multidisciplinary functions and discuss the scientific application of experimental design methodologies as part of the product manufacturing process. Individual topics could include formulation, clinical phases, manufacturing, technology transfer, production scale-up, and optimization.
Production Systems: Articles should focus on the full range and scope of unit operations and production steps for manufacturing APIs, both small molecule and biologic pharmaceuticals, the building and critical process utility systems that support the manufacturing process, as well as the means of managing, dynamically controlling, and automating manufacturing and warehousing operations. Articles could highlight production unit operations, management, and control.
Quality Systems: Articles should focus on the role and elements of a quality management system and its impact on the overall risk management approach, as well as its implementation in a scientific and pragmatic manner. Case studies could demonstrate the purpose, elements, and implementation of risk management and a quality management system and systems validation.
Regulatory Compliance: Articles should highlight regulations and guidance issued by the international regulatory bodies and coalitions. Case studies could demonstrate the application of regulations and industry-generated guidance for global harmonization of compliance and product registration. Topics could include regulations, standards, practices, and guidance related to drugs, the environment, health, and safety.
Research and Development: Articles should illustrate sound research methods and results describing innovative methods and techniques in all aspects of the industry, including manufacturing and applied pharmaceutical science and technology, process and product understanding, and control.
Supply Chain Management: Articles should feature key components and financial impact of supply and distribution chains. Topics focus on the systems required to control and automate the receipt, storage, and dispensation of raw and packaging materials, as well as storage and distribution of finished products. Other topics could include materials management, operational economics, and warehouse and distribution management.
Reader Demographics
Areas of Expertise
Architect/Engineer/Construction: 15.03%
Clinical Materials/Investigational Products: 1.08%
Health/Safety/Environmental: 1.08%
Knowledge Management: 0.73%
Operations/Plant Mechanics(electric, utilities: 9.08%
HVAC)/Facilities/Maintenance Planning: 0.0%
Process Control/Automation: 4.27%
Procurement/Buyer/Planners: 0.19%
Project Management: 8.31%
QA/QC/Regulatory Compliance: 17.43%
Sales and Marketing: 7.79%
Research and Development/Technology: 8.64%
Transfer/Process Development: 0.0%
Statistician: 0.19%
Supply Chain Management/: 0.81%
Warehouse Operations/Logistics Planning: 0.0%
Technical Services/Product Support/: 6.04%
Information Systems: 0.0%
Validation/Qualification/Commissioning: 13.49%
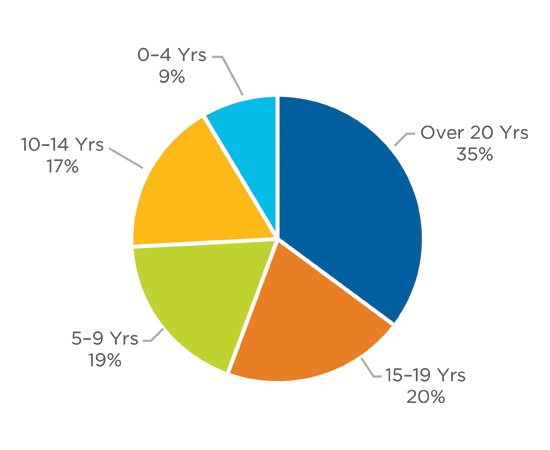
Years in Industry
Additional Resources
Editorial Disclaimers
Letters to the Editor
Pharmaceutical Engineering® welcomes readers’ comments. Letters must include the writer’s full name, address, organization, and years of ISPE membership. If published, letters may be edited for length and clarity. Send correspondence to ISPE Publications Department.
Limitation of Liability
In no event shall ISPE or any of its affiliates, or the officers, directors, employees, members, or agents of each of them, or the authors, be liable for any damages of any kind, including without limitation any special, incidental, indirect, or consequential damages, whether or not advised of the possibility of such damages, and on any theory of liability whatsoever, arising out of or in connection with the use of the information on this site. Learn more about our Terms of Use.
