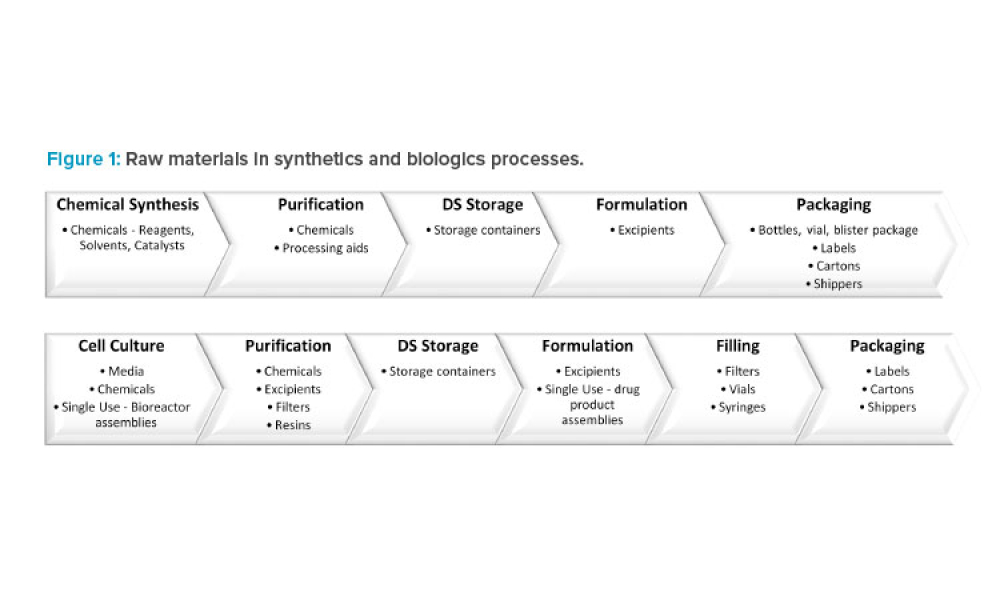
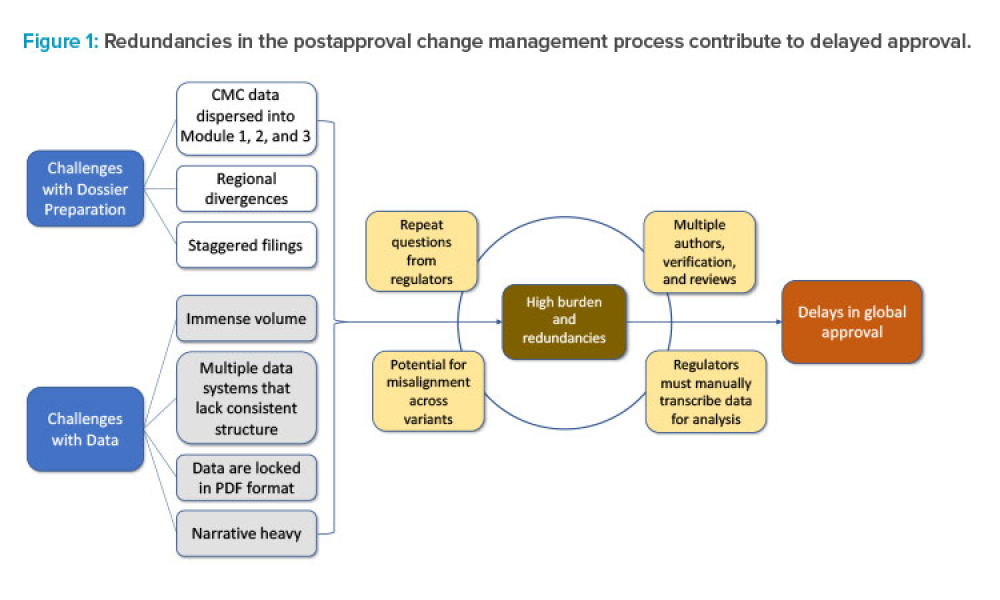
Content highlights regulations and guidance issued by international regulatory bodies and coalitions. Case studies could demonstrate the application of regulations and industry-generated guidance for global harmonization of compliance and product registration. Topics could include regulations, standards, practices, and guidance related to drugs, the environment, health, and safety.
Initiatives
Current Topics
Join the Conversation
As an ISPE member you can engage with 22 active CoPs, including new communities focused on Artificial Intelligence and Sustainability. Connect with experts and join the conversations: Become an ISPE member
ISPE members: Get more involved by volunteering.
Guidance Documents
Active Pharmaceutical Ingredients (2)
+Advanced Manufacturing (1)
+Advanced Therapy Medicinal Products (1)
+Biotechnology (3)
+Critical Utilities (5)
+Drug Shortages (2)
+Good Manufacturing Practice (1)
+Investigational Products (3)
+Knowledge Management (2)
+Lifecycle Management (2)
+Manufacturing Operations (3)
+Microbiological & Viral Contamination Control (6)
+Packaging (1)
+Quality Assurance (2)
+Quality Control (1)
+Regulatory (5)
+Sterile Products (1)
+Supply Chain Management (2)
+Community Discussions
Community Discussions
Aug 11, 2025
Quality
Lifecycle Management
Validation
Aug 06, 2025
Quality Assurance
Jul 25, 2025
Data Integrity
Jul 24, 2025
Information Systems
Artificial Intelligence
Data Integrity
Jul 17, 2025
Manufacturing Operations
Oral Solid Dosage
Jul 08, 2025
Pharma 4.0™
Jun 20, 2025
Sustainable Facilities, HVAC, & Controlled Environments
Pharmaceutical Engineering Magazine Articles
Webinars
Upcoming
On-Demand
ISPE in the News
Latest
-
Validation in Transition: Why 2025 is the Turning Point for the Industry
-
ISPE Webinar: Real-World Trends in GxP including Machine Learning/Artificial Intelligence (Expert Panel Discussion)
-
Machine Learning Predicts Adverse Drug Reactions
-
Prasad Returns to US FDA's CBER
-
Shortage of Sterile Saline Solution in US Subsides
-
Bora Pharma Expands in Minn.
-
Relational Risk Analysis Minimizes Human Error Risk
-
Using Digital Maturity Assessments in Brownfield Facility Upgrades
-
Framework for a Pharma 4.0 Transition
- ISPE Members: Explore Our Volunteer Opportunities
White Papers
September / October 2024
PIC/S in Latin America: Harmonization of cGMP Procedures Cover: This article offers an overview of…
March / April 2024
Navigating the Asia Pacific Pharmaceutical Landscape for Global Impact Cover: The Asia Pacific…
September / October 2023
Regulatory Trends & Quality Initiatives - Shortages of essential medicines around the world have…
January / February 2022
Emerging Leaders has grown from an initiative for interactions among early-career professionals and…

Videos
iSpeak Blog Posts
Featured Conferences