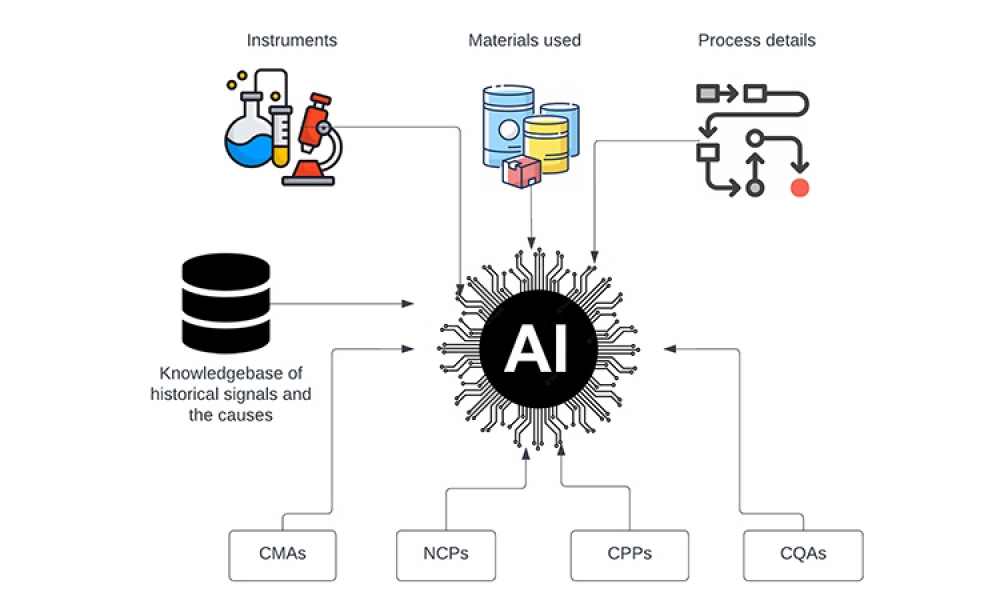
Data Integrity (DI) is part of the mission to ensure the safety, efficacy, and quality of products produced by the pharmaceutical industry, the global regulators (e.g., the US FDA, EMA, etc.) expect that all data submitted by manufacturers to obtain market approval is both reliable and accurate. Regulators consider the integrity of data, from the moment it is generated to the end of its life cycle, to be a critical component of ensuring that only high-quality and safe drugs are manufactured. Data integrity requires that all data be attributable, legible, contemporaneous, original, accurate, complete, consistent, enduring, and available.
Join the Conversation
As an ISPE member you can engage with 22 active CoPs, including new communities focused on Artificial Intelligence and Sustainability. Connect with experts and join the conversations: Become an ISPE member
ISPE members: Get more involved by volunteering.
Guidance Documents
Advanced Manufacturing (1)
+Artificial Intelligence (1)
+Commissioning & Qualification (1)
+Data Integrity (18)
+GAMP® (16)
+Knowledge Management (1)
+Manufacturing Operations (1)
+Pharma 4.0™ (1)
+Quality Assurance (1)
+Regulatory (1)
+Validation (12)
+Community Discussions
Community Discussions
Webinars
iSpeak Blog Posts
Pharmaceutical Engineering Magazine Articles
Videos
White Papers

Pharmaceutical Job Board
Professional Development Training
A GAMP® Guide to Computerized Systems Compliance
+GAMP® Data Integrity 21 CFR Part 11, 2-Day Training Course
+GAMP® Data Integrity 21 CFR Part 11, 3-Day Training Course
+Featured Conferences