A Pharma 4.0™ Future for Brownfield Challenges

Pharmaceutical companies can use digital maturity assessments to address the challenges of upgrading brownfield facilities and implementing digital transformation improvements. Enabled by stakeholder workshops, these assessments can rapidly produce concrete plans and priorities to guide a facility’s development over the next three to five years—delivering business value and laying the foundation for continuous improvement.
The Pharma 4.0™ Necessity
In just a few short years, Pharma 4.0™ principles have evolved for many companies from ‘let’s consider planning’ to ‘why isn’t this done yet?’ Introducing new technologies and processes to stay competitive often places pressure on multiple areas of pharmaceutical operations. Facilities must not only keep pace with one another but also with partners across the value chain—from drug development to materials and packaging.
Meanwhile, competitors may be acquiring advantages that cannot be ignored. Perhaps now is the time: last year’s ISPE Pharma 4.0™ Conference featured multiple case studies highlighting digital transformations,1 and this year’s conference, which is in December in Barcelona, Spain, is expected to have even more. Across our industry, leaders are signaling an increased urgency for change,2, 3 as legacy systems lack the flexibility and efficiency to keep up with digitally paced business in 2025. Nowhere is this challenge clearer, and more relevant, than in brownfield facility developments.
This creates the contradiction: the facilities that could benefit most from Pharma 4.0™ may also be the toughest or riskiest to transform. How do you move forward when there are so many interconnected options, each with implications for your products, processes, infrastructure, compliance, and people?
The Brownfield Contradiction
When planning a new, industry-leading greenfield site, you often have the luxury of embracing multiple aspects of Pharma 4.0™, making cutting-edge innovation a hallmark of the facility. But such opportunities can be rare—more often, development and production must continue at existing sites. Brownfield facilities present unique challenges when it comes to digital transformation.4 Many were built decades ago and have undergone multiple expansions and ownership changes.
Data is a particular concern. Any facility with a history of more than a few years will have a myriad of equipment, systems, and data management practices—resulting in varying levels of data quality and accessibility. There will be data silos, legacy systems, and manual processes that are all running on aging infrastructure. No matter how much you might want to integrate AI analytics, predictive maintenance, or efficiency mapping, is your data even in a form that could use it? Although a guiding framework like FAIR (Findable, Accessible, Interoperable, and Reusable) can offer end goals and guiding principles,5 they can sometimes feel far removed from the reality of an already constructed and operating brownfield facility.
Implementing digital technologies and launching successful digitalization projects is challenging even under the best of circumstances. Research indicates that most organizations may not achieve their transformation objectives—and those that do often struggle to sustain their successes.6 Moving forward requires a way to uncover these challenges and a structured approach to addressing them.
Digital Maturity Assessments
Faced with the myriad challenges and potential solutions in a Pharma 4.0™ upgrade effort, the first best step is a digital maturity assessment. This ensures business value for each recommendation, and drives concrete, measurable business decisions to deliver that value. A successful assessment will cover opportunities across the business—from control strategies to quality management down to the foundational process of an interconnected facility.
Technology only succeeds when the people using it are empowered. That’s why change management—helping both workers and managers become comfortable with new skills and processes—is vital to a successful digital transition.
Why Do a Digital Maturity Assessment?
A 2021 McKinsey study found that among successful digital transformation projects, one of the three leading indicators of successful value capture was in conducting a thorough, evidence-based assessment of the business to identify opportunities for potential improvement.6
We’ve seen more facilities turning to digital maturity assessments not only to get started but also to consistently and successfully drive innovation. A digital maturity assessment can drive decision-making and ensure that progress is made and measured by:
- Preventing business impact from getting lost in technology adoption concerns
- Accelerating the process—assessments only take a few days
- Identifying quick wins and the initiatives that will drive the greatest benefits
- Benchmarking against other manufacturers in the life sciences industry
- Evaluating people and processes alongside technology
Conducting a comprehensive, fact-based assessment of the business can help identify opportunities for improvement. The more thoroughly that an organization uses facts to assess the maximum financial benefit it can achieve from a transformation in the first place, the more confidence leaders will have in setting and pursuing ambitious yet realistic targets that reflect the transformation’s full potential. Additionally, non-financial impacts can be evaluated, ensuring strategic transformation is evaluated alongside return on investment.
Conducting a comprehensive, fact-based assessment of the business can help identify opportunities for improvement.
Standardized Digital Maturity Assessments
Although assessments can be carried out informally or developed in house, we’ve found that using standardized third-party assessment tools offers far greater benefits. They cover a wide range of categories, meaning that they can look at what you’re doing now, but also what you’ll address in the future, making it easier to track progress. These assessments build on the insights of many experts, with valuable considerations built in from other organizations, operations, and industries. They also make it much easier and more valuable to benchmark your organization against competitors and to partner using standardized data. Finally, they offer concrete steps for improvement.
The purpose of this assessment is to understand where digital transformation can deliver business value. Let’s look at the three leading digital transformation assessments that we’ve seen deliver significant value for brownfield pharmaceutical facilities. Although each model has its own slightly different definitions, rating system, and areas of focus, in each assessment you’ll find familiar terms that connect with the realities you see today, and the futures you envision for your facility:
- Low maturity: initial, predigital, manual, paper, ad-hoc, top-down
- Medium maturity: developmental, digital silos, semi-automatic, unit-level automation, labor intensive data retrieval, defined, automatic, digital, interoperable, FAIR, empowered teams
- Advanced maturity: managed, predictive, periodic course-correction, simulated, agile, optimized, adaptive, automatically integrated feedback, real-time adjustments
Option 1: ISPE Pharma 4.0™ Maturity Model and Assessment Tool
Built on the knowledge from its 2023 industry survey on the barriers to innovation that limit the development and implementation of innovative technologies, ISPE released its Pharma 4.0TM Maturity Model and Assessment Tool7 as an attachment to the Baseline Guide. Its goal is to provide practical guidance embedded with regulatory best practices to accelerate Pharma 4.0™ transformations.
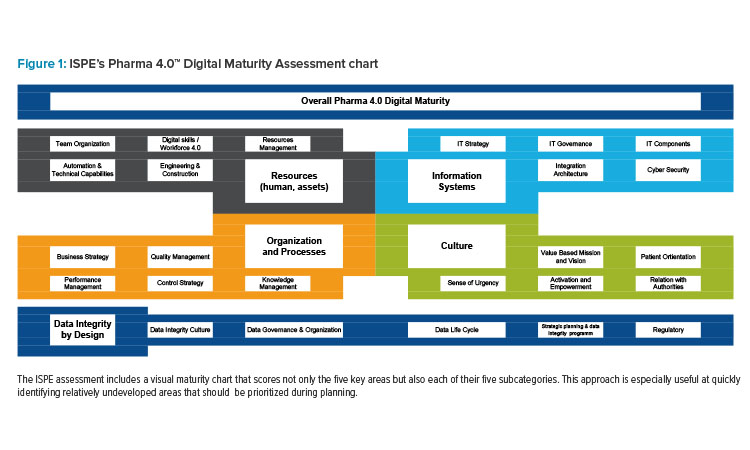
The ISPE maturity assessment looks at five key areas (each with five subcategories):
- Resources
- Information Systems
- Culture
- Organization and Processes
- Data Integrity by Design
Implementing concepts from Pharma 4.0™ requires coordination of these areas, aligning expectations, communication, analysis, and global pharmaceutical regulations. As a result, the ISPE assessment guidelines recommend that the areas of evaluation should have relatively even scores to achieve optimal business results. Increasing only one dimension could have diminished benefits, and a dramatically lagging area could hold back progress in every other area. Balancing these scores then becomes the first priority for planning but also allows for a sophisticated strategy that accounts for each key area of the business.
The ISPE Digital Maturity Assessment is based on the Pharma 4.0™ Operating Model. Its focus on assessing data integrity, for example, has contributed to the growing popularity of digital maturity assessments small-molecule manufacturing. These tools allow organizations involved in the pharmaceutical product lifecycle to fully leverage the potential of digitalization—enabling faster therapeutic innovations and improved production processes for the benefit of patients, while also meeting unique regulatory requirements.
In a 2023 report, McKinsey stated, “In many companies, manual data processes persist, making any advanced analytics at scale or speed impossible. Before pharmaceutical companies can achieve digital and analytics transformation, they must embrace data transformation.”8
We’ve seen the same thing firsthand: data integrity and quality frequently emerge as major inhibitors of digital transformation in the pharmaceutical industry. These challenges often surface during application reviews and are compounded by gaps in harmonized regulations. To address this, ISPE designed the Pharma 4.0™ Assessment as a guided questionnaire to be completed as a group. Having a facilitator present ensures that definitions are aligned between stakeholders and that a shared consensus is developed.
Option 2: BioPhorum Digital Plant Maturity Model (DPMM) Assessment Tool
BioPhorum is an organization that facilitates initiatives across key areas affecting the global biopharmaceutical industry. Its DPMM Assessment Tool9 provides a structured method for assessing a plant’s digital maturity and setting targets aligned with a company’s digital strategy. It can be used for internal and external benchmarking, building technology and investment roadmaps, and defining dependencies across facility operations. While the DPMM and ISPE assessments share similar goals and scopes, each has its own strengths and organizational structure.
The DPMM multidimensional model has eight business and capability dimensions:
- Manufacturing execution
- Process development
- Quality control labs
- Quality management systems
- Manufacturing support
- Supply chain
- People
- Cybersecurity and operations
Scores in each of these dimensions indicate where a facility falls within a five-step digital maturity scale. In the latest version, BioPhorum has increased its own focus on quality control and management systems and has a revised cybersecurity section that focuses on systems that store and transmit digital information. We’ve found this assessment to be especially popular in the biotech sector.
Like the ISPE assessment, the BioPhorum assessment is designed as a questionnaire to be completed as a group. During the assessment, it’s important to ask not only “Where are we at now?” but also “Where do we want to be in the future?” This forward-looking perspective helps gauge the relative importance of each capability and drives prioritization of the gaps to address after the assessment.
Option 3: International Center for Industrial Transformation (INCIT) Smart Industry Readiness Index (SIRI)
The Global SIRI Initiative was developed by the Singapore Economic Development Bank (EDB) and supported by the World Economic Forum.10 It brings together more than 600 manufacturers, governments, and industry associations across 30 countries to accelerate the adoption of Industry 4.0 benchmarking and transformation.11 This global effort has resulted in a highly stable standard that draws on cross-industry data to identify the areas with the greatest impact on cost, industry benchmarks, and KPIs selected by the plants.
Figure 2: A BioPhorum Digital Plant Maturity Model (DPMM) blank chart
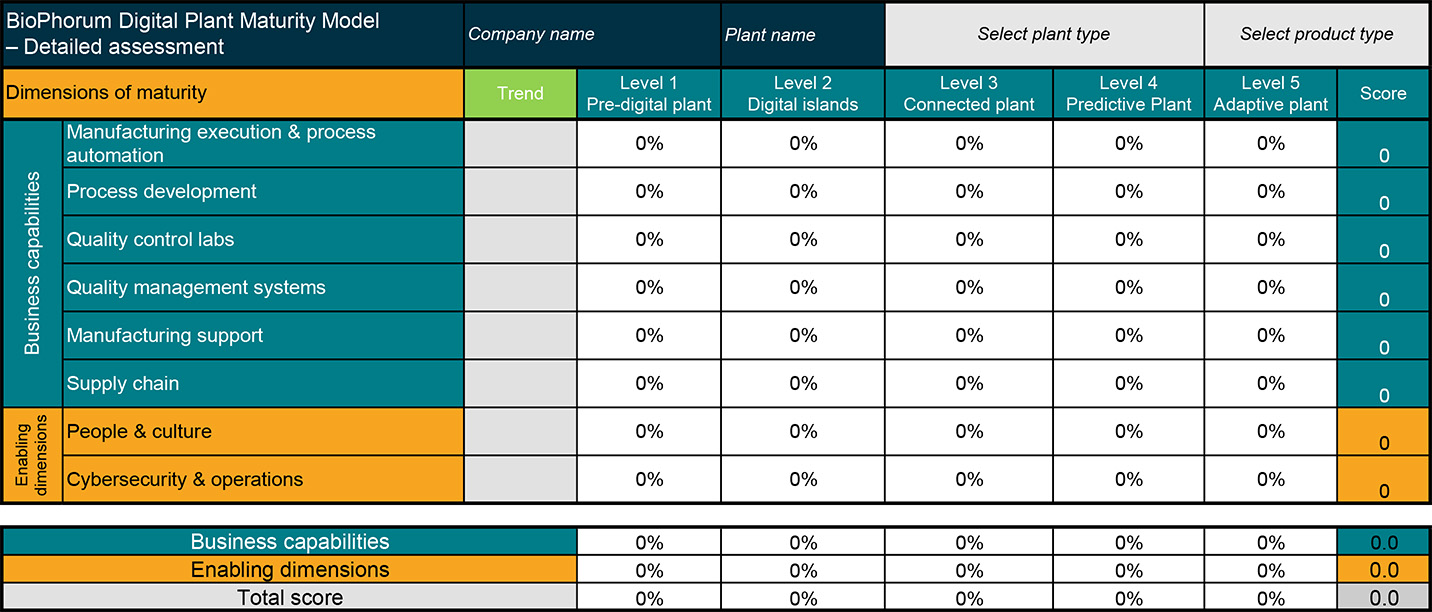
The BioPhorum Maturity Model scores facilities across eight dimensions to calculate a total maturity score. This score is then mapped to one of fi ve progressively advanced levels: predigital plant, digital silos, connected plant, predictive plant, and the highest level, adaptive plant
Figure 3: Example output from a Smart Industry Readiness Index (SIRI) assessment
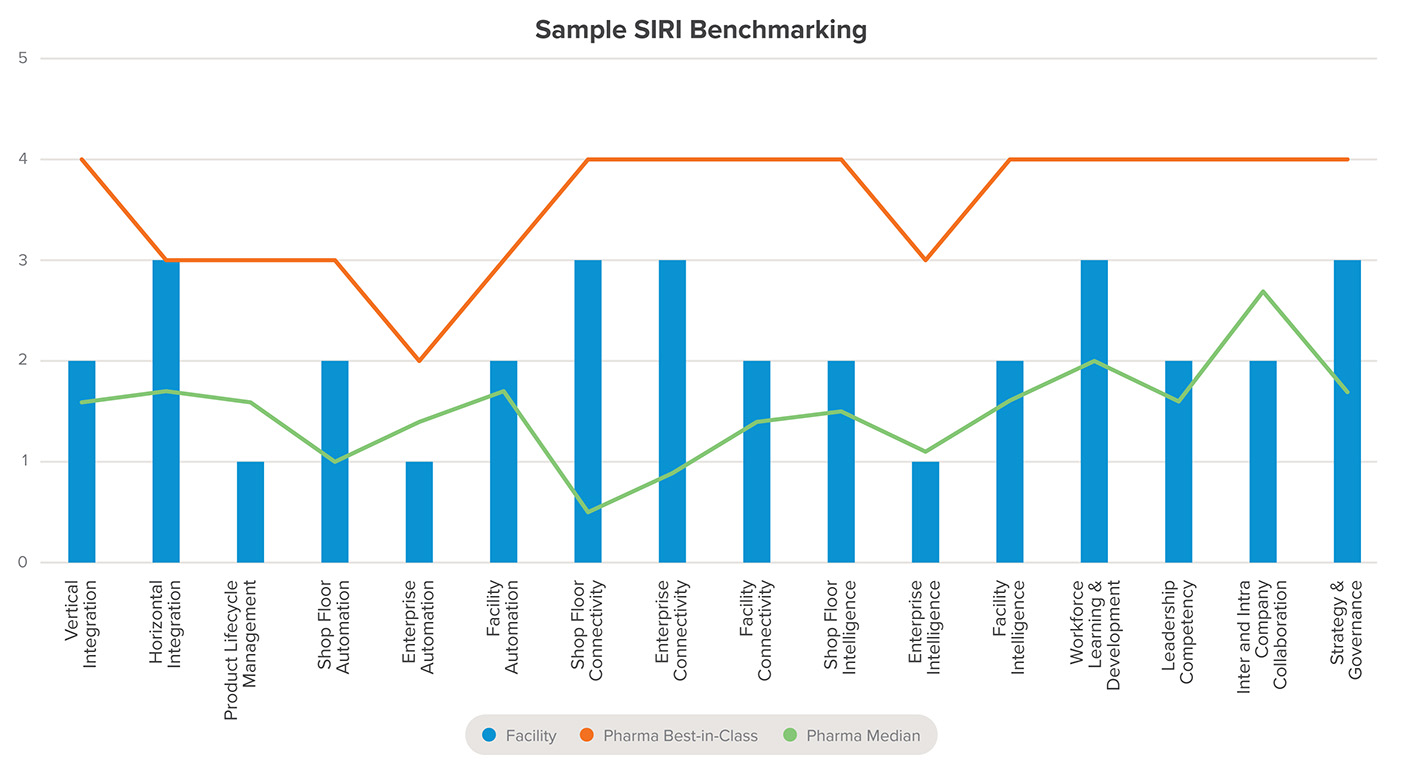
This is a generic example of the visual maturity benchmarking chart from a SIRI assessment. The assessed facility’s maturity scores are mapped against both the industry median and a Best in Class benchmark across all 16 dimensions.
The SIRI Assessment: Two Key Components
- Assessment: The facility is evaluated across 16 dimensions, categorized by organization, technology, and process. Each of dimension receives a maturity rating from Band 0 (the lowest) to Band 5 (the highest).
- Prioritization: The assessment results are combined with additional company-specific information to generate a formal report. This report helps organizations begin—and sustain—their transformation journeys.
This is a generic example of the visual maturity benchmarking chart from a SIRI assessment. The assessed facility’s maturity scores are mapped both to the industry median and a Best in Class standard across all 16 dimensions.
SIRI is fundamentally different from the other two assessments—it doesn’t cover crucial pharmaceutical industry-specific topics like regulatory requirements or data integrity. Instead, it provides helpful benchmarks, KPIs, recommended focus dimensions, and potential areas of impact. A SIRI assessment offers a unique opportunity for any manufacturer to understand their progress, competitiveness, and priorities relative to peers through insightful, industry-agnostic approach—and conducted with a SIRI Certified Assessor. When combined with an assessment expressly built for the needs of the biopharmaceutical industry—and the ability to select KPIs that align with your organization’s priorities—SIRI becomes an enormously powerful. One of the biggest conversation drivers after an assessment is facility performance against benchmarks.
The Maturity Assessment Process
Once you know the assessment(s) you want to use, it’s time to plan the workshop. This is a high-value, low-impact process that can take a few days. Using external facilitators will help set standards across facilities and projects, ensuring a consistent approach that looks similar to the following process:
Pre-Workshop
Prior to the workshop, conduct a questionnaire for all participants.
Workshop
- Review questionnaire results
- Define goals and objectives
- Select business drivers for the facility
- Identify and discuss pain points
- Discuss ongoing projects
- Tour the facility and talk to operations
- Score according to assessment criteria
- Review and align based on the maturity assessment
It’s important to begin the process by observing participants and understanding their needs. Adopting new technology works is most effective when it applies to specific pain points. Ask: What do we need to solve? What improvements can be implemented? Pharma 4.0™ solutions span many areas—manufacturing, filling, packaging, warehousing, quality, maintenance, and IT. Starting with the solution, rather than the problem, will only distract and lead to diminished value in implementations.
Secrets to Successful Assessments
Prioritize Workshop Collaboration
Time and again, we’ve seen that the most important aspect of this entire process is the workshop itself. Every voice is at the table—from production to compliance, and from HR to IT— working through questions, digging into challenges, and discussing what the most successful solution would look like. This one-of-a-kind workshop not only helps everyone understand the project, but also ensures that all participants are aligned and committed to the process. Even if a workshop lasts only a day or two, its impact can extend much further. A data transformation effort may take months, or even years, and the benefits can pay off for decades. One of the most impactful parts of the assessment process is getting everyone with different perspectives in the same room. That’s often when the real breakthroughs happen and shared understanding begins to take shape.
Shift Mindsets and Operations
It is critical to remember that digital transformation is about more than just adopting new digital technologies—it’s a shift in mindset and in how companies operate to deliver value for patients and customers. As shown in initial ISPE research, the “fear of change” is real.12 Digital transformation requires not only capabilities but also a new adaptable way of thinking.
The standardized, benchmarked point of view that an assessment provides makes conversations with stakeholders and executives much easier. While decision-making around digital transformation initiatives in our industry remains under-researched,13 we’ve seen firsthand that investing, or not investing, in digital technology projects becomes clearer with the insight and context provided by an assessment.
Document Commitments and Insights
Successful assessments benefit from visible tracking, which ensures clear communication and empowered action, with all parties understanding their shared responsibility for change. This kind of documentation can protect the investment in the whole process. We’ve seen the best results occur from workshop participants capturing commitments on visual boards, lab leaders tracking communication about progress, and analysts empowered to introduce external data and experiences.
Combine Digital Assessments for Multiple Perspectives
Each maturity model has its own strengths and weaknesses, making it more difficult to determine in advance which one will provide the most insights for your facility— especially if you’re just getting started. Since coordinating calendars is often one of the most difficult challenges in conducting an assessment, consider performing multiple assessments simultaneously in the same workshop. In practice, it will only add an incremental amount of work and can deliver more value and even unexpected insights. For example, data integrity insights often align with the vertical integration dimension from the SIRI model, offering a broader and more strategic picture. As you continue to update assessments to track progress, it may not be necessary to carry multiple assessments forward. However, after completing the full process once, you’ll likely gain more confidence in the value each model provides.
Even after completing these assessments, the next steps may still be unclear. For example:
- The ISPE assessment may show a relatively low score in Data Integrity by Design.
- The BioPhorum DPMM might indicate a gap between the current and ideal states of manufacturing execution systems.
- The SIRI assessment could recommend focusing on vertical integration, shop floor intelligence, shop floor connectivity, and strategy and governance.
So, what should be the real priority? In this scenario, it’s helpful to pause and examine the underlying reasons behind each model’s recommendations. Ask: What KPIs are they aligned with? What business capabilities would you gain by advancing this maturity? This will allow you to build internal alignment, allowing your team to acknowledge the most critical gaps and develop a plan to get there.
Figure 4: Example Pharma 4.0™ roadmap schedule
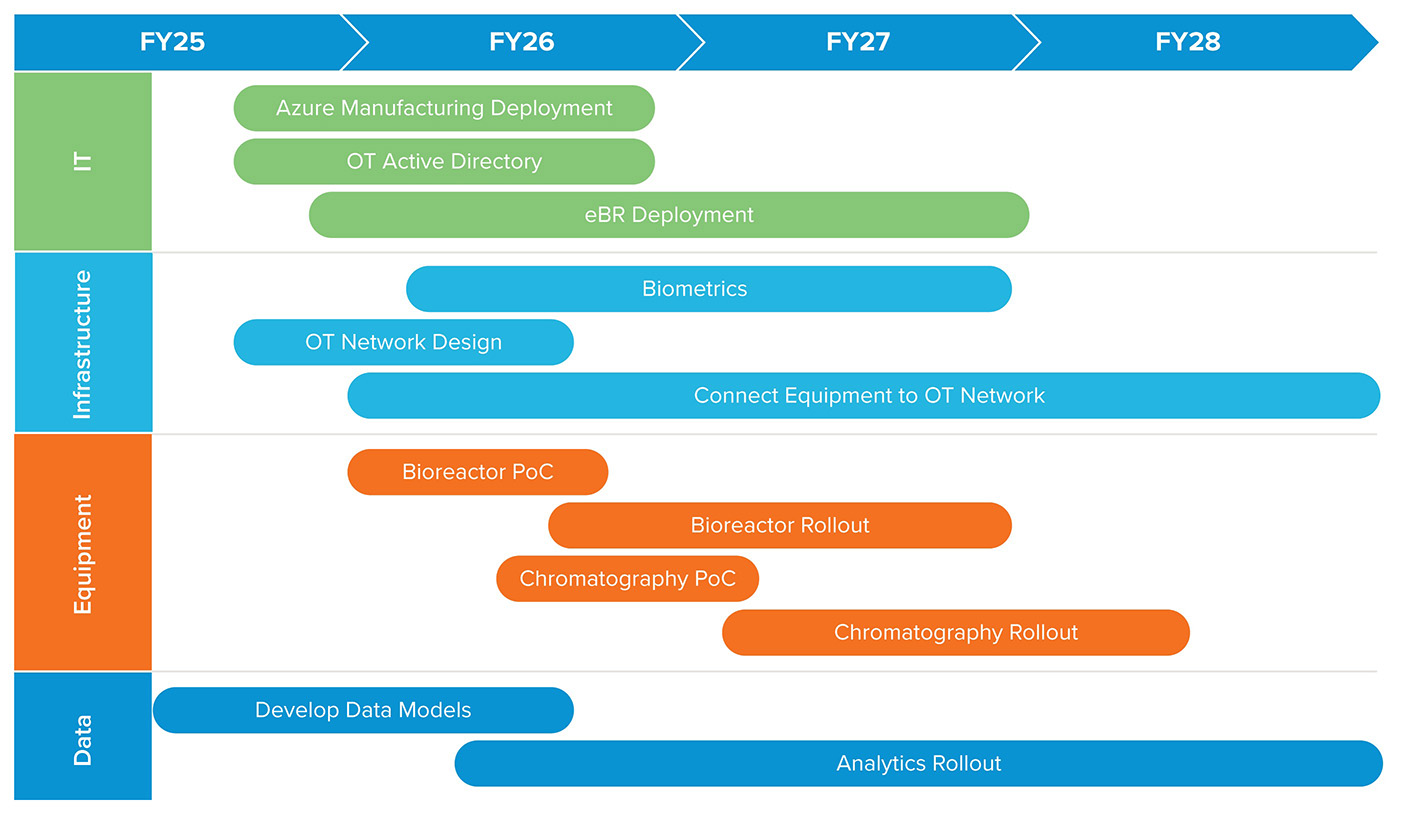
This example of a Pharma 4.0™ development roadmap shows how insights from a digital transformation maturity assessment can inform planning and resource allocation over a multi-year timeline.
From there, you can begin identifying the technology and process changes needed to close those gaps and build business cases to support them. This could involve:
- Exploring solutions already in use within your manufacturing network.
- Conducting external benchmarking using resources like the ISPE Baseline ® Guide Vol 8: Pharma 4.0™ or the World Economic Forum Digital Lighthouses.
- Engaging with solution providers to drive conversations and align on the impacts to your technology and operations landscape.
Next Steps: Driving Business Decisions
This is where many organizations falter. Without taking steps to quantify and ensure business value as their project scales, initiatives often becomes stuck in the pilot stage. Our goal is to equip stakeholders with a practical framework to digitally transform operations—securing a competitive advantage in an industry driven by rapid innovation and regulatory demands.
The initial assessment is followed by several steps to deeply quantify the benefits of proposed changes and develop a detailed digital roadmap. For example, a successful assessment should enable capital expenditures planning for the next 3–5 years.
Technology Deep Dives
This process involves mapping improvement opportunities identified during the assessment to current market offerings (if available), developing capital cost profiles, calculating return on investment, and identifying any non-financial benefits aligned with the goals outlined in the initial assessment.
These deep dives are essential to building a business case for a digital transformation project that every stakeholder will understand.
In the previous example, the recommendation to enhance both data integrity and vertical integration requires identifying solutions that address both aspects. For instance, if paper records pose data integrity risks, this presents an opportunity to enhance vertical integration by ensuring data interoperability across all necessary systems. This approach mitigates data integrity risks associated with manual entry, such as transcription errors, lack of data validation, and delayed error detection. As a bonus, it also generates valuable data that can improve operations and performance in the future.
Roadmap Development and Maintenance
Deep dives into each proposed technological development can inform a sophisticated, multi-stage roadmap to guide your Pharma 4.0™ progress over the next three to five years.
A digital roadmap includes:
- Desired capabilities and technologies
- Key enablers, vendors, and infrastructure
- Business impacts (ROI / NPV, KPI, etc.)
- Pilot opportunities and roll-out strategies
The SIRI Prioritization Matrix helps make this possible by balancing cost, KPIs, and facility assessment matrix scores to bring focus to the efforts that will yield the greatest benefits. There are always areas for improvement with any plan. It’s worth identifying these as you go, drill down to fully identify the opportunities, and then adopt the appropriate technologies to solve new problems. We recommend conducting annual assessments to track progress and maintain momentum in your digital transformation efforts.
Even though the ceiling of opportunity for digital transformations is very high, the practical reality is that the floor can be quite low.
The Future: Leadership and Adaptation
Digital transformation is an exercise in leadership and continual improvement. The humbling power of connected manufacturing lies in its ability to highlight weaknesses and reveal opportunities to innovate and improve. As such, consistent reevaluation and openness to changing the ecosystem you’ve put in place are critical to long-term success.
Even though the ceiling of opportunity for digital transformations is very high, the practical reality is that the floor can be quite low. As we have observed in numerous assessments, digital transformation in pharmaceutical facilities frequently begins with a bold, confident first step from paper to digital records. According to CRB’s 2022 Horizons: Life Sciences report, only 27% of respondents prioritized funding specifically for digitizing supply chain information14 and supply chain is just one component of a much larger picture.
Conclusion
Although there are justifiable reasons why our high-stakes, heavily regulated industry has been reluctant to transition to all-digital records, the benefits of doing so are consistently dramatic, positive, and—in 2025—are proven. Organizations that make this leap immediately gain a deeper understanding of their own operations across departments but also unlock new possibilities. Digital information is more flexible and fungible, it can be analyzed and shared in ways that enhance efficiency, reliability, and flexibility.
In our experience, meaningful cross-departmental initiatives like digitization only succeed when everyone is brought into the same room. Time and again, we’ve seen a maturity assessments act as catalysts for organizational change. In the Horizons: Life Sciences 2022 report by CRB, we found that most of those surveyed considered their companies to be at least at Level 3 of the DPMM, which signifies a connected facility, a high level of automation, and a strong intent to advance further.
This intent represents both an opportunity and a responsibility for leadership. Revisiting progress, celebrating wins, and diagnosing failures keeps teams excited and engaged. Assigning ownership to each technology initiative as its own mini project, can pay dividends in follow-through, accountability, and collaboration. When people can associate faces with an initiative, they tend to respond more positively, helping sustain momentum over the years required for continuous digital transformation.




