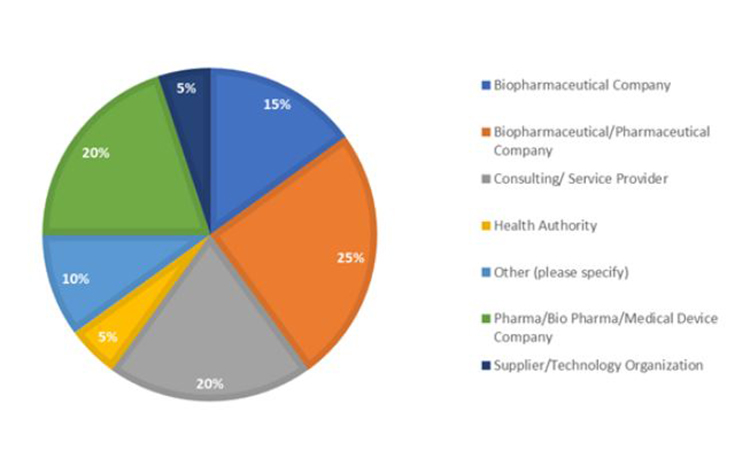
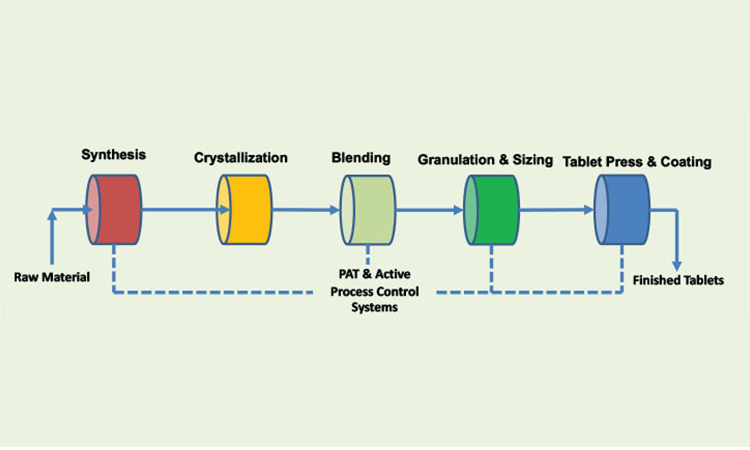
Read, Learn, Innovate: Pharmaceutical Engineering® Summer Reading Part 1
Featured in this edition of the Pharmaceutical Engineering® Online Reading Roundup are suggested summer reading ideas—articles on important topics from recent issues to revisit when you have some down time this summer—or to enjoy for the first time in case you missed them. Learn more about knowledge management implementation, zero carbon, MES, and more.