It’s hard to imagine life without instantaneous weather and time; however, some readers of this blog may know when weather, some years ago, was a mystery, and accurate time required a call from a pay phone to verify the correctness of your new Timex© watch!
Submit Your Best Content to ISPE
ISPE’s official blog, iSpeak accepts contributions from our Members and professionals in the pharma industry.
Featured in this edition of the Pharmaceutical Engineering® Online Reading Roundup are some articles to start off the new year right! We revisit a favorite topic, sustainability—and will be updating our coverage of this topic with the upcoming March-April 2023 issue of Pharmaceutical Engineering.
It has been a long journey from the development of primarily manual aseptic processes to fully automated systems that do not require manual intervention. Looking back in history, it was a step-by-step development forward to the current contamination control strategy (CCS) many decades ago. The risk of contamination of a sterile product from the environment (room) and the people who do work in...
Building off of the success of the 2022 ISPE Aseptic Conference, the program committee has developed a robust program covering key areas in aseptic manufacturing, processing, and filling for students, emerging...
Cultural Excellence is the expressed and implied ways in which an organization operates. Excellence in organizational culture is essential for delivering robust and sustained quality performance and ensuring patient-focused outcomes. Culture determines quality outcomes by affecting the organization’s ability to identify and act upon near-miss shortages, ensuring the integrity of its data,...
ISPE’s Women in Pharma® group strives to tackle unconscious biases that limit women, and other marginalized groups, within the pharmaceutical community. Over the last few years, the group has grown to be a global force across borders, connecting industry professionals through regional and international programming, including having a presence at the 2022 ISPE Annual Meeting &...
Multi-Use Filling Line for RTU Syringes, Vials, Cartridges and Bulk Vials - Photo courtesy of INCOG BioPharma Services
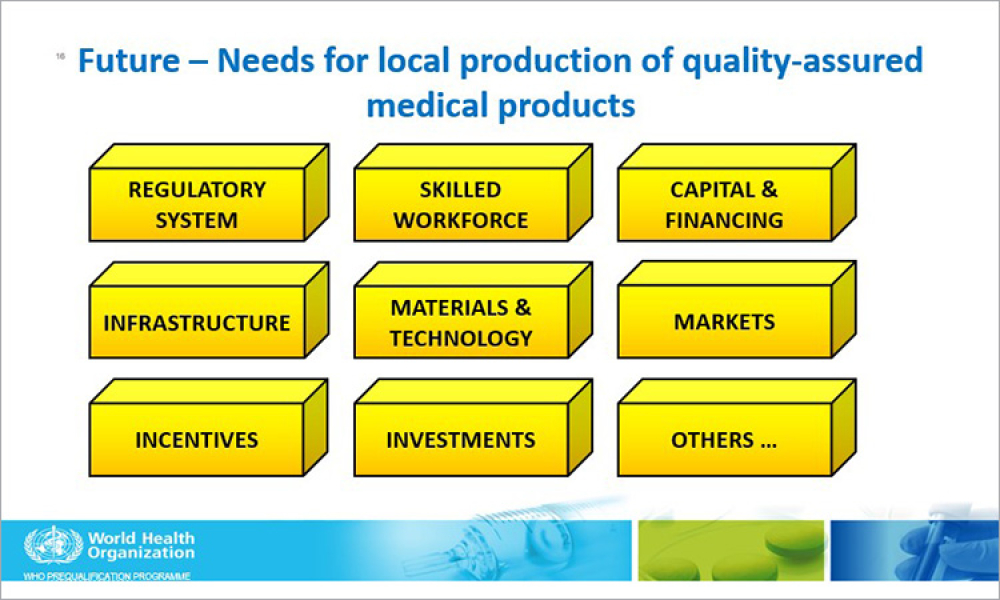
The definition of technology is “the set of knowledge, skills, experience, and techniques through which humans change, transform and use our environment in order to create tools, machines, products and services that meet our needs and desires.” A...
ISPE’s Women in Pharma® ended the year strong with their latest webinar: Digitalization in Business Processes that Support Sustainability & Economy of Means.
We are The Culture Club, a self-formed cross-COP group of individuals who came together to discuss challenges encountered as we transition quality and validation into the digital world and identify ways to influence cultural changes needed within our industry to better enable innovation. As we are not all from the same COP, we first need to establish a way to connect and collaborate with all...
Excellence is everywhere, and it must be illuminated. For the fourth consecutive year, the annual ISPE Italy Affiliate Award for women in the pharmaceutical sector will be organized by the local group of ISPE Women in Pharma® (WIP). This important initiative was created to celebrate the commitment, perseverance, professionalism and competence of the women who work in this sector, qualities...
The ISPE Singapore Affiliate 2022 Conference and Exhibition is a WRAP! I was fortunate to chair both the pre-conference symposium opening plenary and the ‘Post Pandemic Transformation’ track.
A key challenge in life sciences is the detrimental effect of unplanned downtime and lost batches due to unanticipated equipment failure. Traditional maintenance programs are calendar or condition-based and can range from being overly conservative to reactive, leaving money on the table (over-maintenance - “just in case”), or jeopardizing productivity altogether. As described in the ISPE Good...
As one of the founders of Cheme Engineering, Nik Krpan was instrumental in the start-up of the company. He became president of the company in 2012. Nik says that leading the company through continued growth has been an exciting aspect of his career. “We started with five people and now we have more than sixty. It’s been really rewarding to be part of a creating an environment where people can...
The 2023 ISPE Aseptic Conference will devote one entire day to case studies related to aseptic manufacturing projects. For us, seeing the real-life challenges that the industry is facing and how project teams...
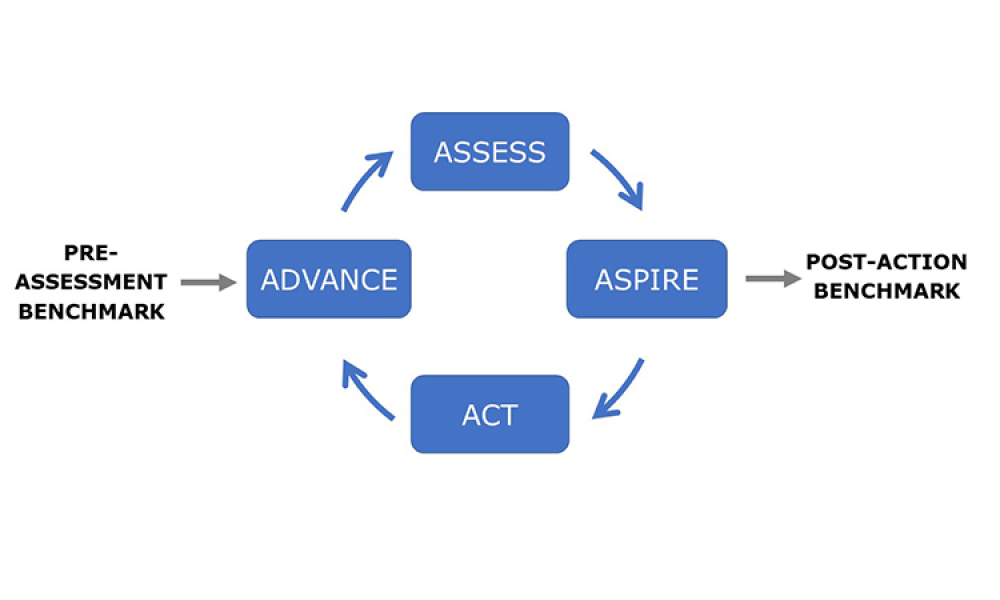
ISPE is pleased to announce the complete Advancing Pharmaceutical Quality Program (APQ) Program is now available with the release of the fifth and final guide.
By the ISPE Regulatory Team
ISPE’s regulatory volunteer groups are charged with bringing visibility and solutions to major challenges faced by the industry with regard to regulatory/quality matters, and facilitating the flow of information between ISPE members and global Health Authorities. Support for that mission was on display at the 2022 ISPE Annual Meeting and...
The Pharmaceutical Inspection Co-operation Scheme (PIC/S) celebrated its 50th anniversary at a special symposium in Dublin, Ireland on 4 October 2022. Since the first meeting of the PIC Committee in 1972, PIC/S has come a long way! The symposium was titled “Thriving at 50 and Striving Forward” and was aimed at promoting and...
As we are all aware, the COVID-19 pandemic has had a unique, world-wide effect on the pharmaceutical industry as well as health authorities. At the height of the pandemic, and continuing as it appears to wane, health authorities have not been able not carry out normal, in-person inspections of pharmaceutical manufacturing sites. However, in the ”Age of Covid,” using the Internet and associated...