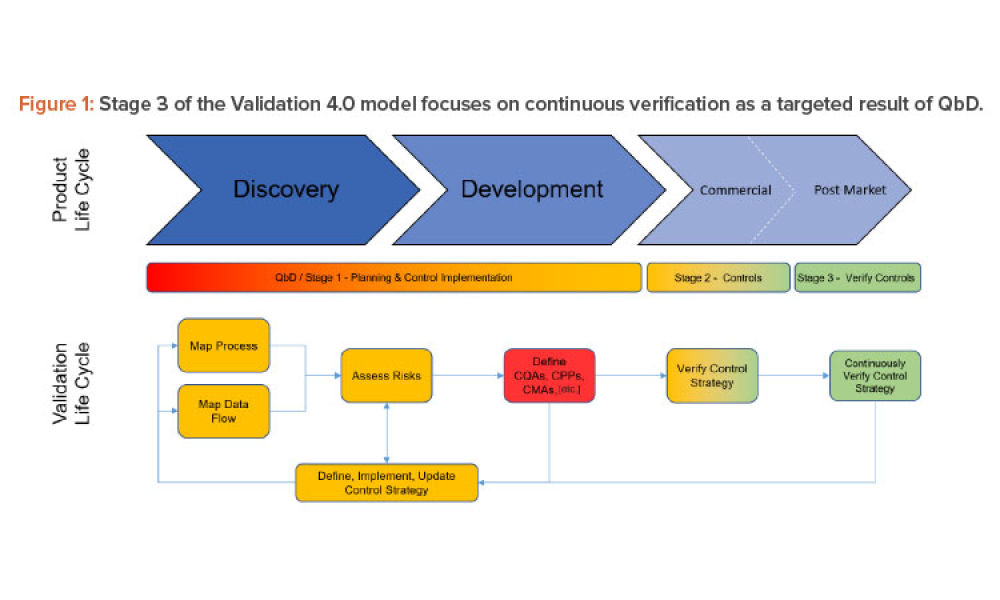
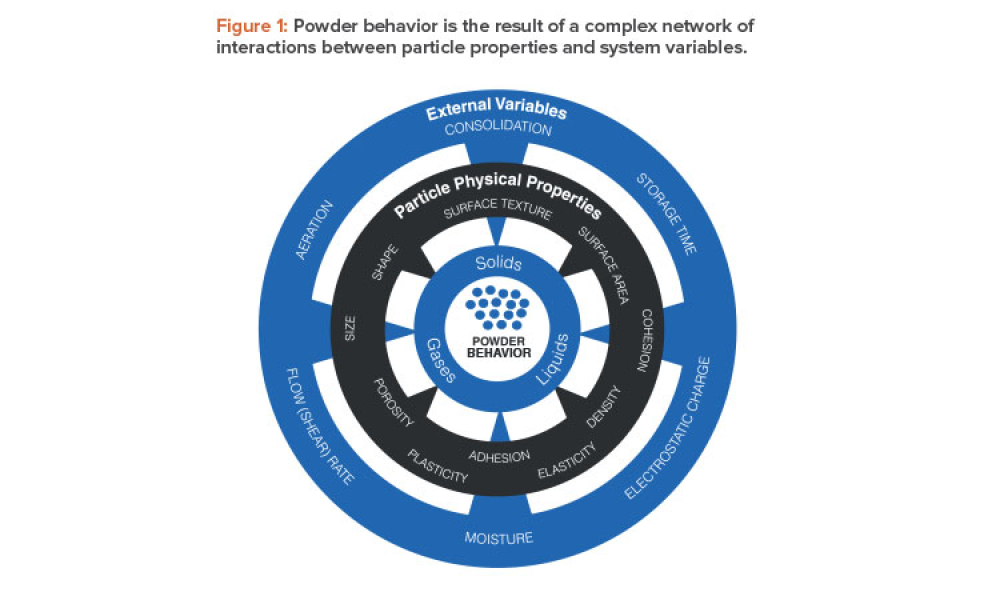
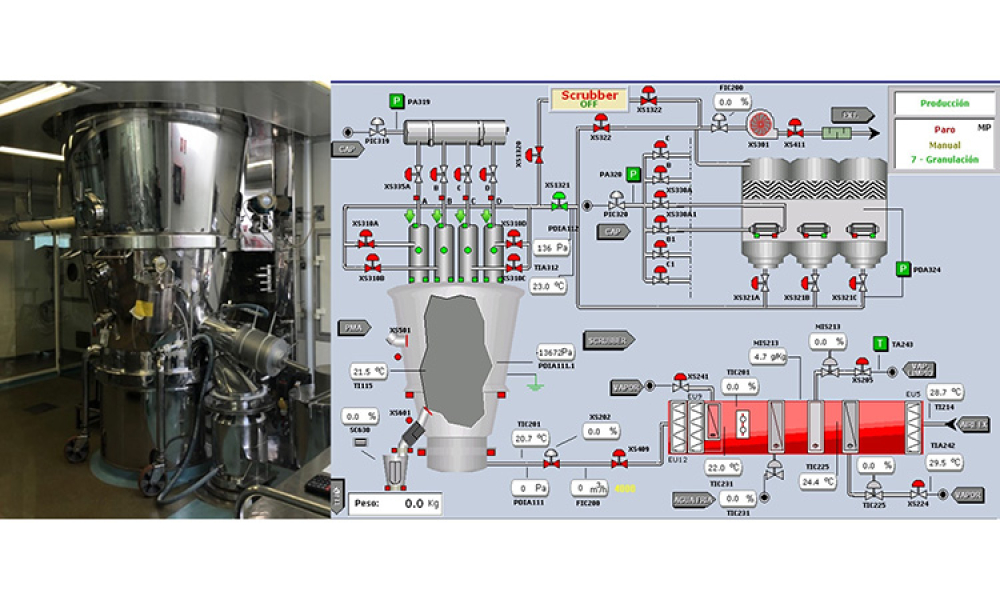
Oral Solid Dosage (OSD) is the inspection and evaluation of the manufacturing and control processes used to manufacture solid oral dosage form pharmaceutical products.
Join the Conversation
As an ISPE member you can engage with 22 active CoPs, including new communities focused on Artificial Intelligence and Sustainability. Connect with experts and join the conversations: Become an ISPE member
ISPE members: Get more involved by volunteering.
Guidance Documents
Advanced Manufacturing (1)
+Manufacturing Operations (2)
+Microbiological & Viral Contamination Control (1)
+Oral Solid Dosage (2)
+Community Discussions
Community Discussions
Jul 17, 2025
Manufacturing Operations
Oral Solid Dosage
Apr 16, 2025
Information Systems
Regulatory
Advanced Manufacturing
Artificial Intelligence
Mar 28, 2025
Information Systems
Regulatory
Advanced Manufacturing
Artificial Intelligence
Nov 04, 2024
Regulatory
Supply Chain
Active Pharmaceutical Ingredients
Good Manufacturing Practice
Oct 31, 2024
Regulatory
Quality
Good Manufacturing Practice
Sustainable Facilities, HVAC, & Controlled Environments
Pharmaceutical Engineering Magazine Articles
White Papers
May / June 2025
The ATMP Issue: In this issue, we focus on the manufacturing of advanced therapy medicinal products…
March / April 2025
A Skill Management Framework for a Pharma 4.0™ Workforce Feature: Pharma 4.0™ is driving fundamental…
January / February 2025
GAMP® is indispensable for safeguarding the safety, quality, and compliance of pharmaceutical…
November / December 2024
Plastic Process Waste in Biopharmaceutical Manufacturing Cover: This article presents a…
September / October 2024
PIC/S in Latin America: Harmonization of cGMP Procedures Cover: This article offers an overview of…

iSpeak Blog Posts
Videos
Professional Development Training
ICH Q6A: Specifications, Test Procedures, and Acceptance Criteria for New Drug Substances and Products
+GMP Sterile Pharmaceutical Manufacturing Facility Training Course
+White Papers
May / June 2025
The ATMP Issue: In this issue, we focus on the manufacturing of advanced therapy medicinal products…
March / April 2025
A Skill Management Framework for a Pharma 4.0™ Workforce Feature: Pharma 4.0™ is driving fundamental…
January / February 2025
GAMP® is indispensable for safeguarding the safety, quality, and compliance of pharmaceutical…