The 2022 ISPE Aseptic Conference Regulatory Panel session on 15 March was as always one of the highlights of the conference, and this year’s discussion with regulators followed in that tradition.
September / October 2022
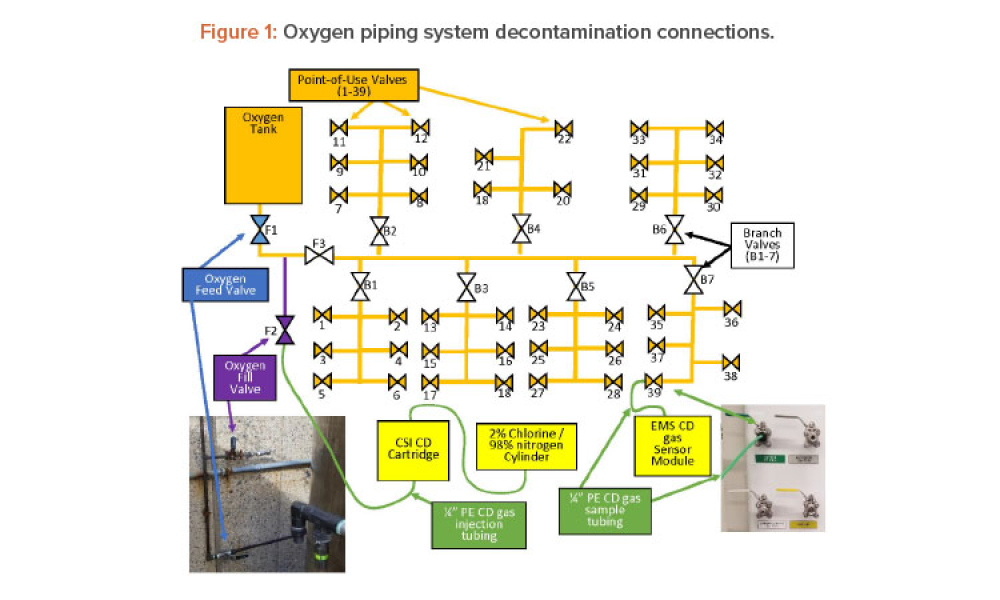
Chlorine dioxide has been shown effective in decontaminating various types of chambers and volumes such as rooms, isolators, processing tanks, and entire facilities, but its use to decontaminate compressed gas piping systems has not been documented. This article discusses using dry gaseous chlorine dioxide (ClO2) to decontaminate an oxygen (O2) feed piping system in a...
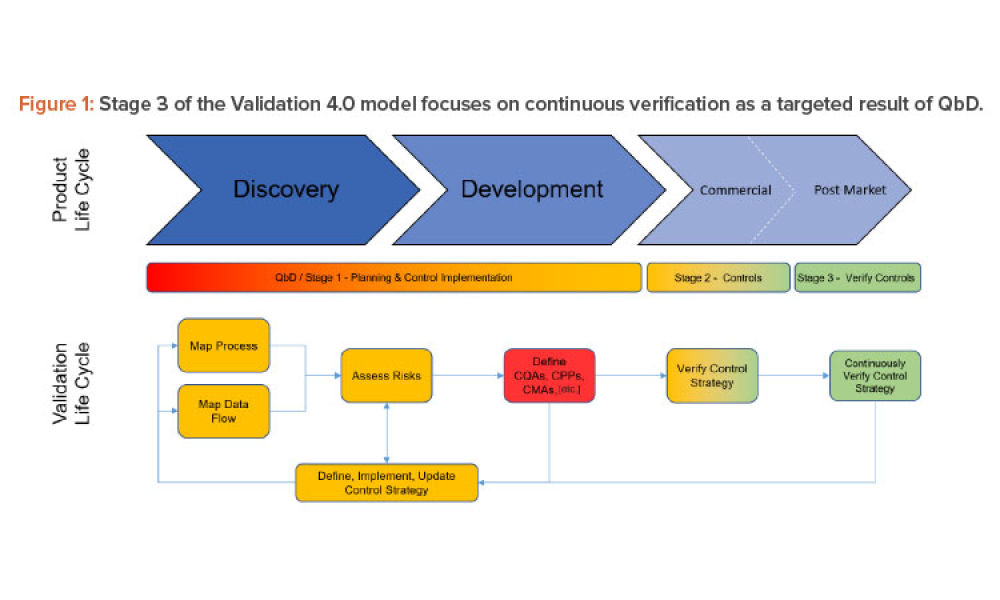
Three case studies on Validation 4.0 demonstrate how quality by design (QbD) principles, when applied with digitization, can verify processes in scale-up and technology transfer, and why blend and content uniformity matter for tablet integrity.
ISPE has more than 19,000 members from more than 129 countries, and many of them spend countless hours sharing their knowledge and connecting with others, helping ISPE to advance the educational and technical efficiency of all members.
Four teams of Emerging Leaders (ELs) represented one of four different company manufacturer types in the Emerging Leader Hackathon at the ISPE 2022 Europe Conference. This article provides some participants’ views of...
The Europe Hackathon for students and recent graduates was held 23–24 April as part of the activities during the 2022 ISPE Europe Annual Conference. This article provides an overview of the event and how it...
The members of the Sterile Products Processing (SPP) Community of Practice (CoP) Steering Committee are as diverse and varied as the topics they discuss. The Community of Practice is comprised of professionals from around...
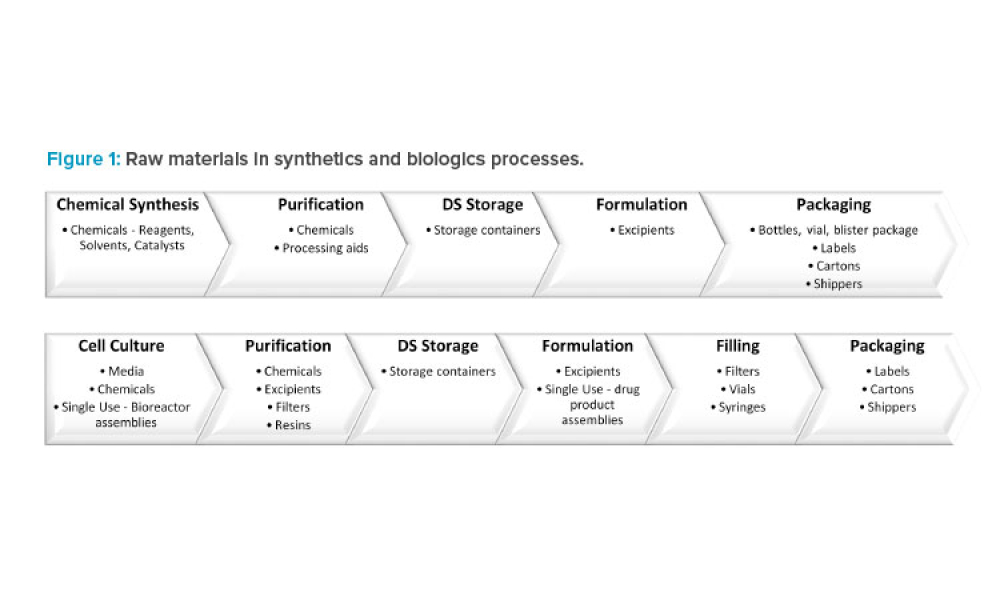
A reliable supply of raw materials is critical to maintain a robust supply chain to serve patients globally. With shortages, regulatory complexity is compounded due to differences in submission and data requirements from various regulatory agencies. Therefore, there is an increasing need to implement a harmonized regulatory infrastructure that is both flexible and predictable to provide more...
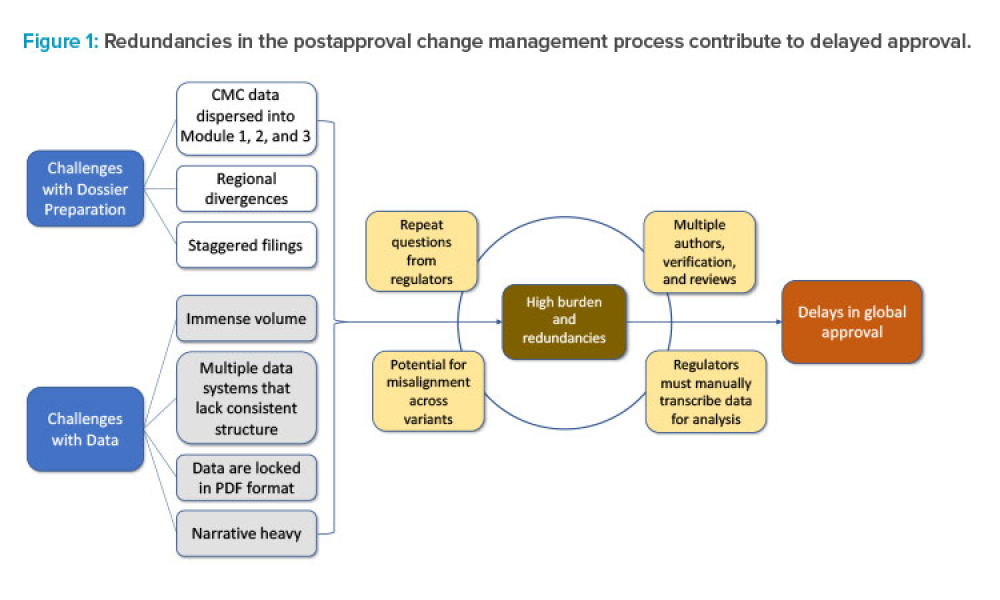
Postapproval change management of pharmaceuticals is an essential part of life-cycle management but is associated with regulatory challenges. Incorporating concepts and tools from the International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) Q12 guideline, combined with structured content and data management (SCDM) and a cloud-based data exchange...
The International Council for Harmonisation of Technical Requirements for Pharmaceuticals for Human Use (ICH) guideline on Registration of Pharmaceuticals for Human Use (M4)