ISPE is committed to fostering communications and interactions to advance common interests among the pharmaceutical industry and regulatory agencies.
Dialogue with Regulators
ICH Q12 Implementation Strategies Discussed During Webinar on Challenges and Success of ICH Q12

Challenges and successes of ICH Q12 lifecycle-related submissions were the topic of a discussion with the USFDA at a February 2021 webinar.
2020 ISPE Annual Meeting & Expo: How Regulators are Responding to COVID-19’s Impact

The Global Regulatory Town Hall held at the 2020 ISPE Annual Meeting featured regulators from ANSM, ANVISA, MHRA, US FDA and the WHO discussing challenges to the agencies during the pandemic, and how resilience and adaptation had increased interaction and convergence among regulators.
Regulatory Panel Addresses COVID-19 Challenges

Regulators from AEMPS, EDQM, MHRA, Russia’s GxP Institute, US FDA and the WHO addressed COVID-19 learnings, digitization, and the value of industry interaction and collaboration for crises management at the ISPE European Annual Conference.
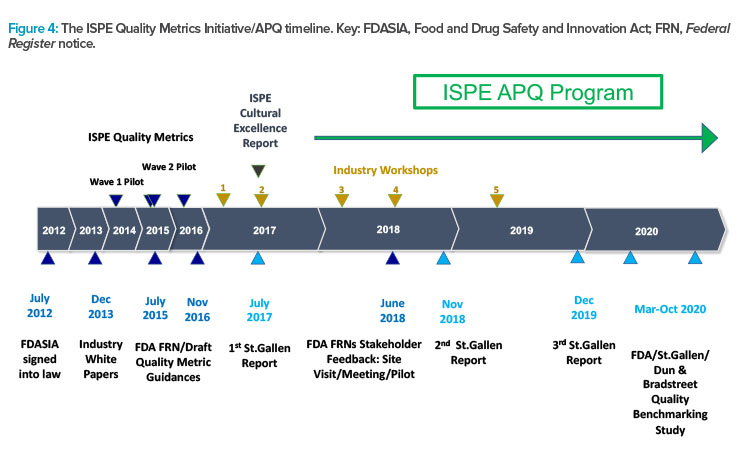
Regulators Discuss Distant Assessments During and After COVID-19
Another panel at the ISPE European Annual Conference featured regulators from AIFA, ANSM, EMA, MHRA, PIC/S, Swissmedic, and Russia’s GxP Institute discussing learnings from the pandemic related to inspection management, the possibilities for using distant assessment of GMP now and in the future, Pharma 4.0, and the practicalities of harmonization.
Singapore Affiliate’s 20th Anniversary Conference

HPRA and HSA Regulators on Industry & Regulator Collaboration
At the ISPE Singapore Affiliate’s 20th Anniversary Conference, HPRA and HSA regulators and a senior representative from industry discussed current and future collaboration between industry and regulatory authorities.

Regulators from HPRA, HSA, MHRA & TGA Discuss Distant Assessments
Also during the Singapore Affiliate’s 20th Anniversary Conference, regulators from HPRA, HSA, MHRA ad TGA responded to questions on the future of distant assessments.
ISPE Singapore Affiliate Interviews HSA’s Sia Chong Hock
2021 ISPE Global Pharmaceutical Regulatory Summit
Remote/Distant Assessments, Audits, and Regulatory Guidance
ISPE will host a virtual summit on Remote/Distant Assessments, Audits and Regulatory Guidance on 28 April 2021 with the participation of AEMPS, ANSM, MHRA, PIC/S, Regierungspräsidium Darmstadt, Russia SID & GP, US FDA, and WHO. Attendees will be able to submit questions for the regulators in advance of the Summit.
FDA Commissioner on Lessons learned from COVID-19
In a presentation during the 2020 ISPE Annual Meeting & Expo, former FDA Commissioner Stephen M. Hahn, MD, spoke about building upon the lessons learned from COVID-19 to speed the advancement of new ideas and practices.
ISPE Participated in DAF ACT Initiative to Support Domestic Manufacturing of API
.jpg)
ISPE recently participated in the U.S. Department of the Air Force Acquisition COVID-19 Task Force (DAF ACT) to advise Regulatory, Technical, and Workforce elements favorable to creating a more robust and sustainable domestic pharmaceutical manufacturing base for Active Pharmaceutical Ingredients (APIs). A report was issued.
ISPE’s Advancing Pharmaceutical Quality Program Launched
ISPE launched the Advancing Pharmaceutical Quality (APQ) Program with the publication of the ISPE APQ Guide: Corrective Action and Preventive Action (CAPA) System. The guide introduces the Assess, Aspire, Act, and Advance framework to determine the maturity of a firm’s CAPA system as an important element of a Pharmaceutical Quality System. The guide is the first in a planned four-part series that seeks to improve the state of pharmaceutical quality and ensure sustainable compliance.
Drug Shortages – Looking Forward
Drug Shortages: Looking Forward with Regulator Insight

In two forums at ISPE 2020 European and Annual Meetings regulators from several health authorities (AEMPS, AIFA, ANSM, FDA, MHRA, and WHO, and FDA), emphasized the importance of health authority engagement and business continuity planning.
Business Continuity Planning to Prevent Drug Shortages
Business continuity planning is a powerful, methodology, essential for enabling an uninterrupted supply of critical medicines for patients in challenging manufacturing circumstances. ISPE’s Drug Shortages team published a “30,000-foot view” article outlining principles and best practices of business continuity planning to ensure continued pharmaceutical supply.
ANVISA training facilitated by ISPE’s Latin America Regulatory Group
The Latin America regional group of ISPE’s Regulatory and Quality Harmonization Committee (RQHC) facilitated three days of customized training for 34 ANVISA assessors in late 2020. The courses were customized versions of ISPE’s Process Validation and Combinations Products training courses tailored to ANVISA’s specific interests and delivered by ISPE training instructors.
ISPE’s Regulatory Committees, Working Groups, and Initiatives
ISPE’s volunteer-led committees, working groups and initiative team are driving ISPE’s efforts to deliver operational excellence and regulatory insights to enhance industry efforts to develop, manufacture and reliably deliver quality medicines to patients.
Regulatory Resources:
- Regulatory Initiatives and Groups
- Regulatory Commenting
- Regulatory Updates
- Regulatory & Quality Networking Community (Exclusive Member Benefit)
For more information, contact Carol Winfield, ISPE Sr. Director Regulatory Operations