In honor of the 40th birthday of ISPE and Pharmaceutical Engineering®, we looked back to 2010 to see what technology-focused articles were published in the magazine. Here are just a few:
September / October 2020
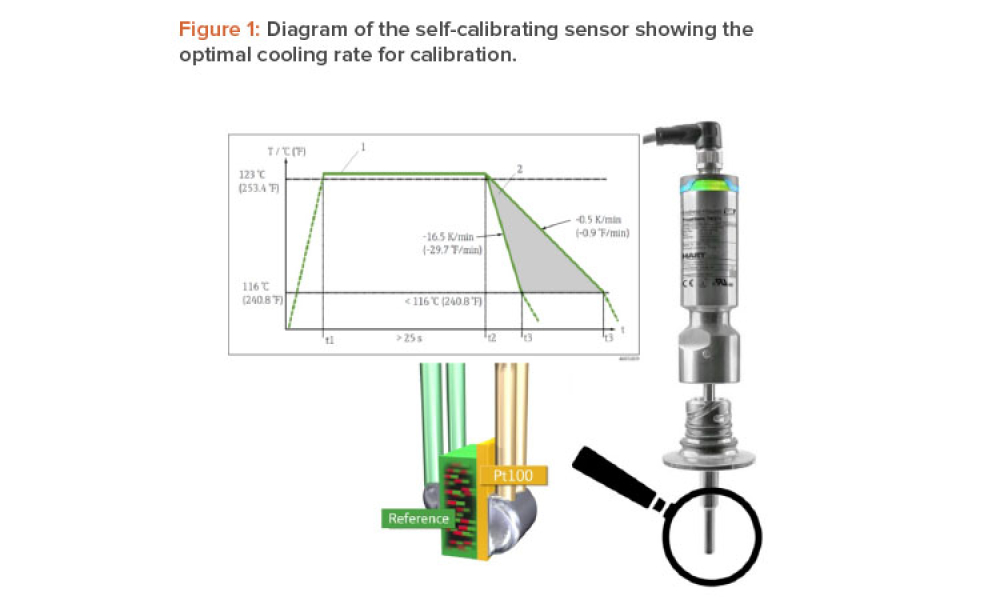
A temperature sensor in a medical autoclave is typically calibrated once a year. If the sensor proves to be inaccurate, all batches produced since the last calibration must be evaluated. Endress+Hauser has developed a self-calibrating sensor that automatically verifies its accuracy during each sterilization batch. This article describes a case study at the Merck Healthcare KGaA sterile...
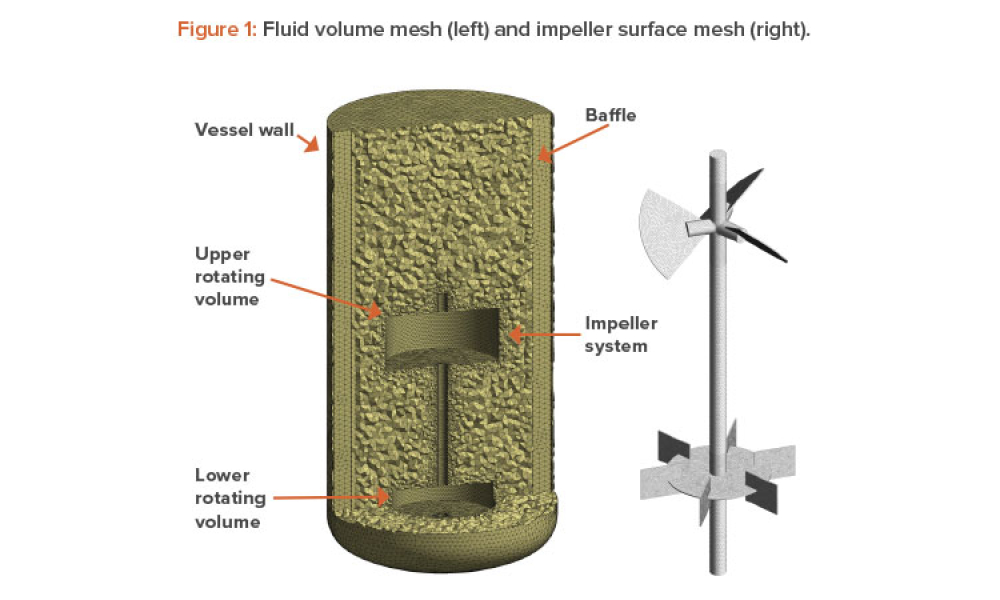
Computational fluid dynamics (CFD) can reduce or eliminate the need to perform bioreactor scale-up studies because full-scale manufacturing bioreactors can be simulated to predict performance. This article discusses the use of computational fluid dynamics for that purpose, to predict the performance of a manufacturing-scale bioreactor under various operating conditions.
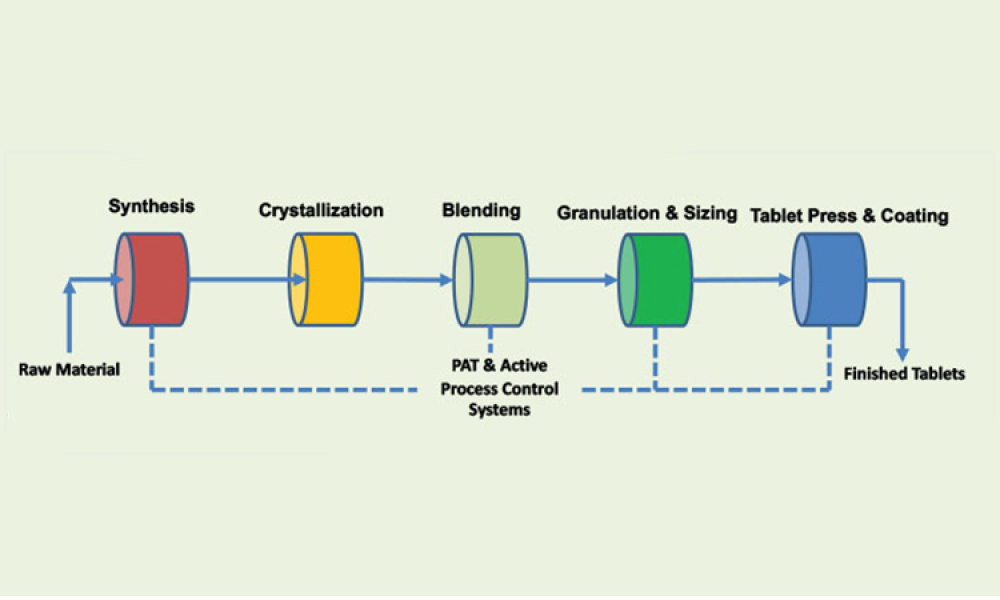
Process analytical technology (PAT) is perceived as the main enabler for a robust control strategy with continuous manufacturing (CM) because process analytical technology can aid in implementing continuous manufacturing throughout the entire life cycle. This article discusses quality and regulatory hurdles in the life cycle of a process analytical technology application—including model...
ISPE has published a new Good Practice Guide: Critical Utilities GMP Compliance—How to Be Compliant and Ready to Prove It. Written and reviewed by...
In March 2020, the ISPE Eurasian Economic Union (EAEU) Affiliate was officially established. This event was a significant step along the journey to integrate members from Eurasian Economic Union nations into the international pharmaceutical community, as well as another sign of progress in the development of a harmonized scientific,...
Each year, ISPE celebrates innovations and advances in pharmaceutical manufacturing technology with its Facility of the Year Awards (FOYA) program. This year, we added a new category, Social Impact, to recognize companies that developed new standards and practices to prevent drug shortages and increase patients’ access to medicine, designed new tools or techniques that reduced the cost of drug...
The 2020 ISPE Annual Meeting & Expo will be ISPE’s first completely virtual Annual Meeting. As always, there will be great learning and networking opportunities—in fact, the digital format offers greater flexibility for attendees.
The ISPE France Affiliate is fortunate in many ways. The pharmaceutical industry in France is world class, employing close to 100,000 people and generating €55.9 billion in annual revenue.