The 30th ISPE Aseptic Conference culminated in the interactive regulatory panel, which offered attendees the unique opportunity to ask questions directly to the regulators.
May / June 2021
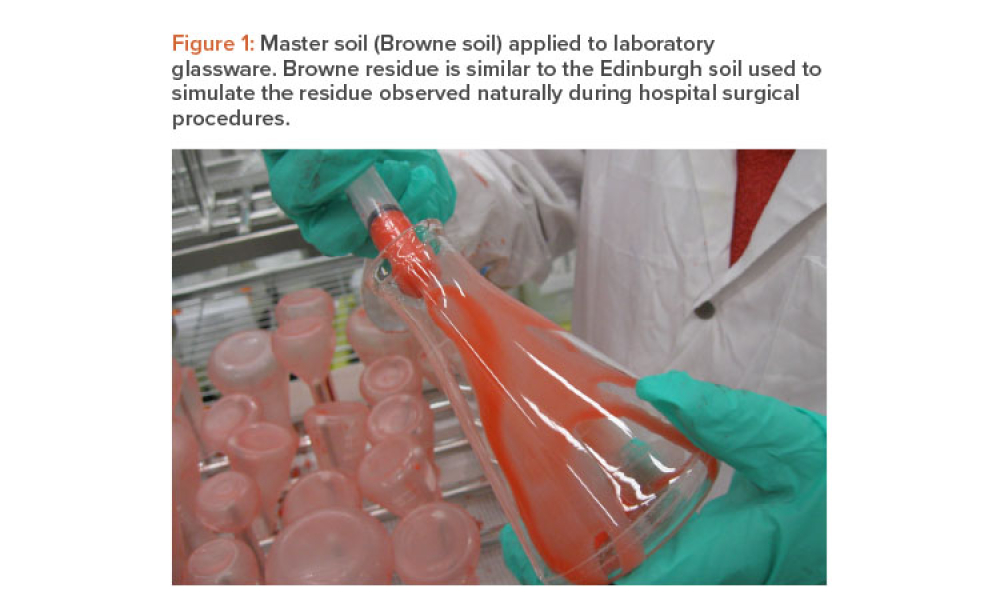
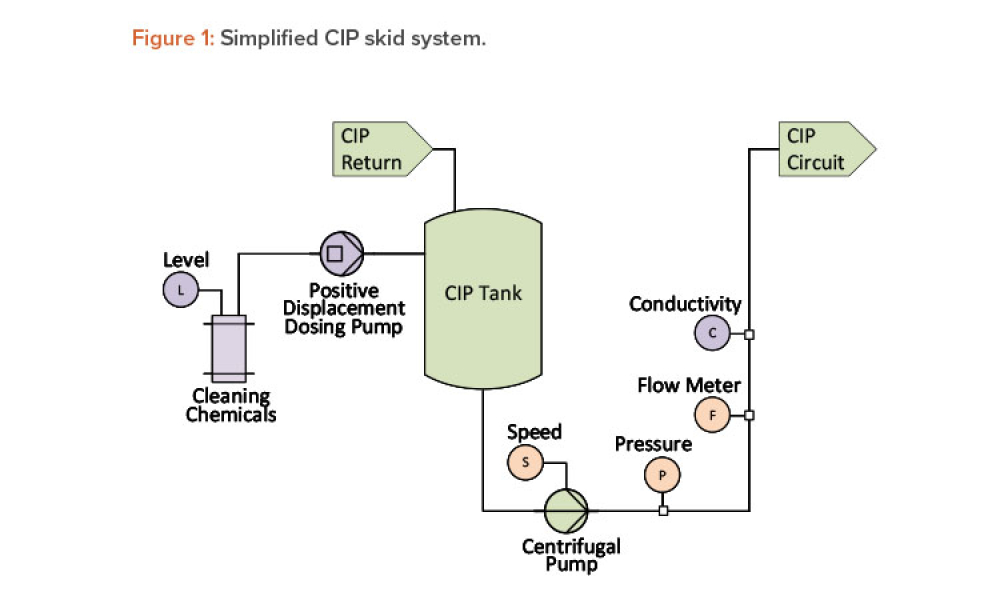
One of the goals of the cleaning validation design phase is to define critical process parameters (inputs) and acceptance criteria (outputs) of the cleaning process. This article explores the selection of a master soil as part of the cleaning validation design phase for automated parts washers. The selection and qualification of a master soil through laboratory testing and during factory...
What if the reliability of a system could be improved by accessing the standard data provided with modern process instrumentation? These data, accessed from existing instrumentation, can be used to analyze the fitness of processes, equipment, and instruments; better understand processes; support discrepancy investigations; and provide a data-driven basis for the timing of maintenance and...
Maintenance can impact both the quality of products and the compliance of pharmaceutical processes, and maintenance programs have long been recognized as critical to the success of the operations they support.
ISPE Emerging Leaders (ELs) held the first fully virtual International Hackathon in February. Fifty-one participants from over 22 countries encountered real-life challenges with working remotely and across time zones. Innovation...
This second of a two-part series explores digital transformation and digitalization in the biopharmaceutical industry with information about how data science enables digitalization along the product life cycle. (Part 1 was published in the March-April 2021 issue of Pharmaceutical Engineering.
This article provides a beginner’s overview of how organizations can achieve a state of preparedness (readiness) for inspections, with a specific focus on IT systems.
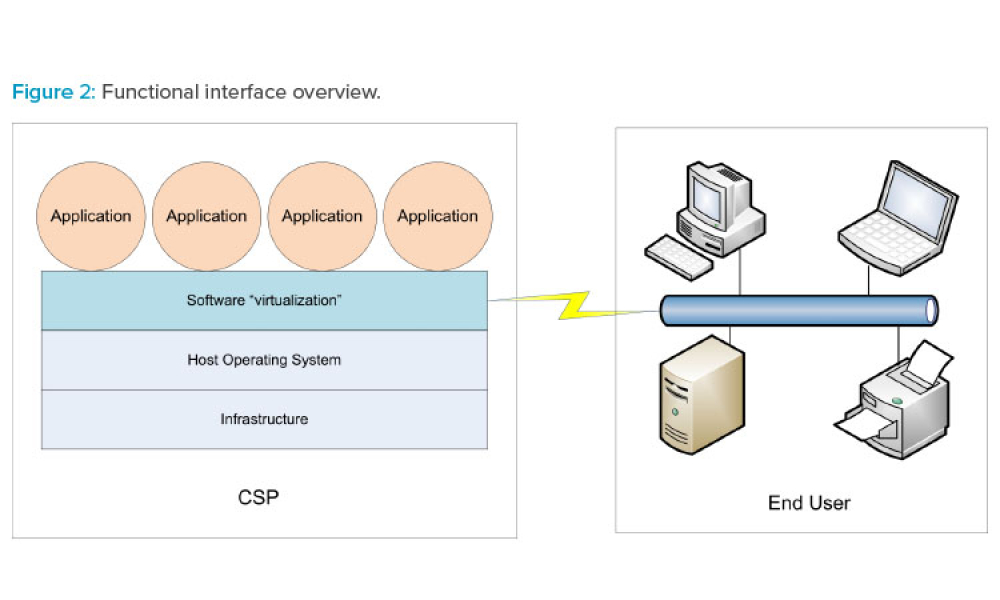
Cloud computing can be described as networked access and utilization of configurable computing resources such as data and information storage, processing capabilities, applications, and other services on computerized systems provided and/or maintained by a remote organization. As life sciences companies consider the advantages and costs of utilizing cloud services, they first need to invest...
In 2021, the ISPE GAMP® Community of Practice (CoP) is celebrating 30 years of promoting industry good practice for computerized systems and encouraging technical innovation and progress, while protecting patient...
Real-world evidence (RWE) is clinical evidence regarding the usage and potential benefits or risks of a medical product derived from analysis of real-world data (RWD) relating to patient health status and the healthcare delivery.