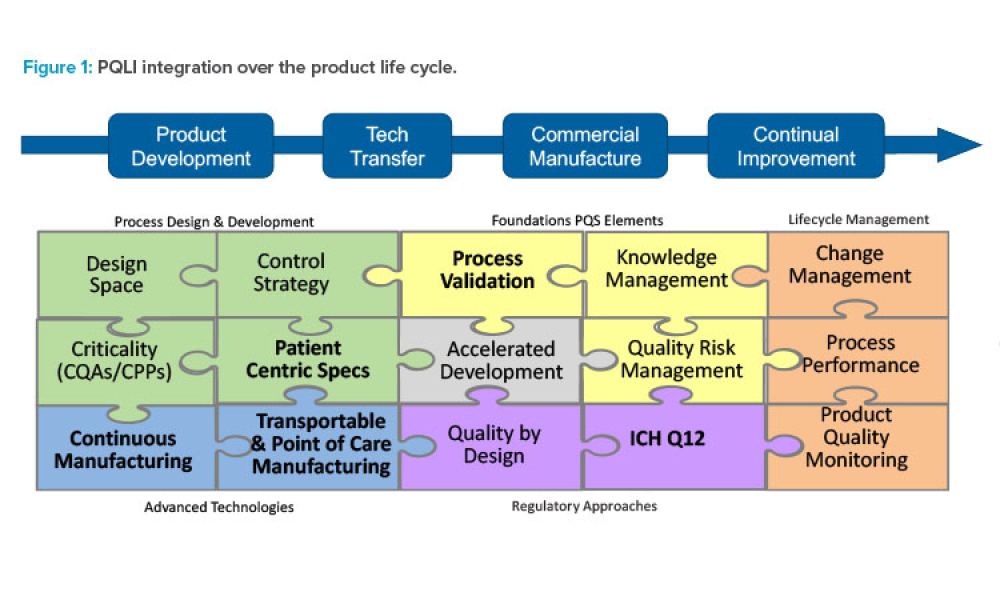
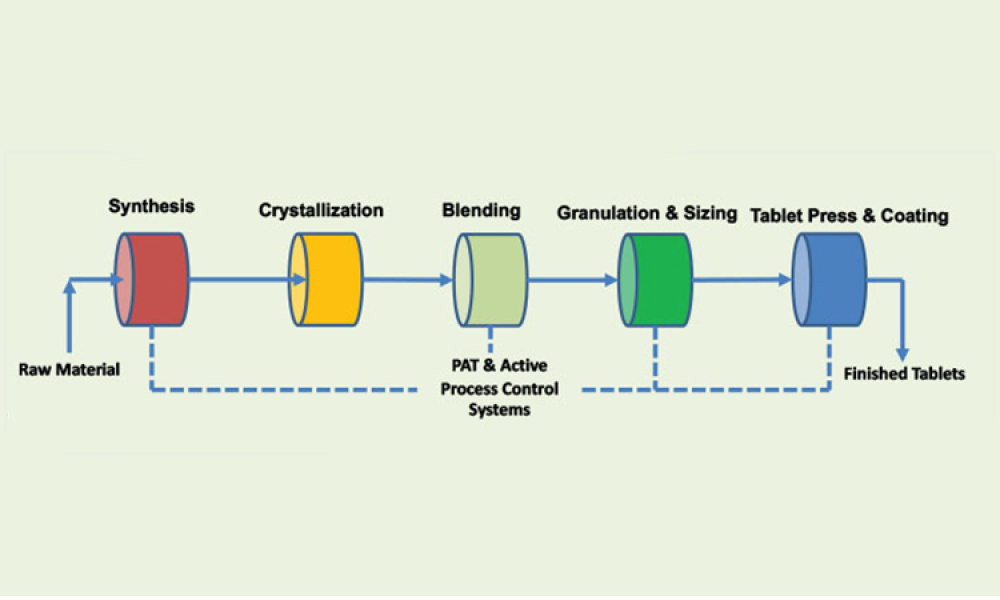
Features
Gabriella Dahlgren, PhD

Johnson & Johnson Innovative Medicine
Director, Strategy Deployment and Excellence, PQM
Gabriella Dahlgren, PhD, oversees a team that manages stability for the Janssen SM portfolio. She has been part of the Global Janssen Product Quality organization since 2015, initially focused on leading the development of new procedures and processes to support PAT, real-time release, and CM. She is currently Co-Lead of the ISPE PQLI® Continuous Manufacturing team. Prior to joining Janssen, Gabriella’s experiences included more than 10 years as an analytical chemist in various functions, focused on end-to-end process creation for method development, test method validation, new technology eval-uation to speed up release testing, and hydrophilic coating implementation on new medical devices. She also served as the cross-functional subject matter expert for resolution of production issues as they relate to testing and chemical characterization. She has a PhD in bioanalytical chemistry from the University of Michigan and a BS in chemistry from Southeastern Louisiana University. She has been an ISPE member since 2015.