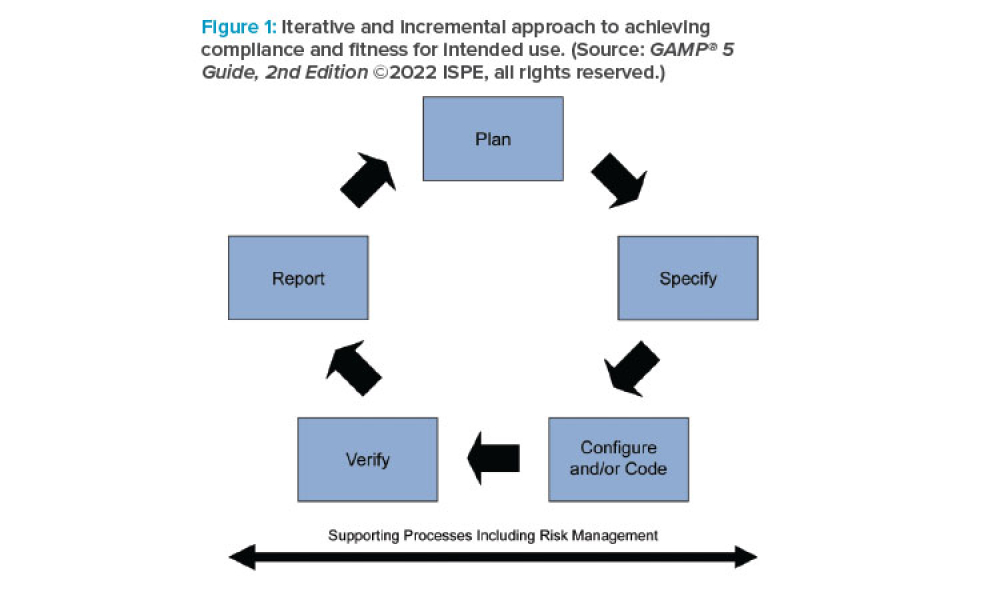
In the pharmaceutical industry, ensuring the safety, quality, and efficacy of products is paramount. With the increasing complexity of manufacturing processes, the growing reliance on computerized systems and advancements in technology, GAMP® guidance provides a framework for validating and maintaining the integrity of computerized systems, ensuring they are fit for their intended...
Sion Wyn

Related Articles
Risk assessment is essential, however, risk assessment is of limited value if it is not conducted by a team with the necessary process, product, and functional understanding. Conducting risk assessments prematurely may lead to invalid assessment of overall risks. Conducting risk assessments too late will limit the opportunity to address design flaws and effectively test processes and...
This article describes a practical and pragmatic approach to the management of computerized system life cycle and information technology (IT) process records. The objective is to effectively achieve and maintain compliant GxP-regulated systems that are fit for intended use, and to support patient safety, product quality, and data integrity.
Computer software assurance (CSA) has been discussed widely in industry over the past five years. While the principles are well understood and welcomed, until now some of the practical detail on how exactly to implement CSA into an organization has been missing.
ISPE’s GAMP® 5: A Risk-Based Approach to Compliant GxP Computerized Systems (Second Edition) (GAMP® 5 Guide, 2nd Edition) maintains the principles and framework of the first edition and updates their application in the modern world, including the increased importance of service providers, evolving approaches to software development, and expanded use of software tools and...
This article explores life-cycle activities for machine learning (ML) within regulated life sciences. It positions and contextualizes the life cycle and management of the machine learning subsystem or components within a wider system life cycle. It also gives general descriptions and guidance illustrated by a case study demonstrating a machine learning application to medical image recognition,...