When seeking a monitoring system, you are confronted with an abundance of choice. The options are significantly reduced when the monitoring system is for a GxP application, as most system vendors are simply not equipped to provide the requisite level of detail and control needed to satisfy internal quality and external regulatory requirements. Even still, there is sufficient choice available...
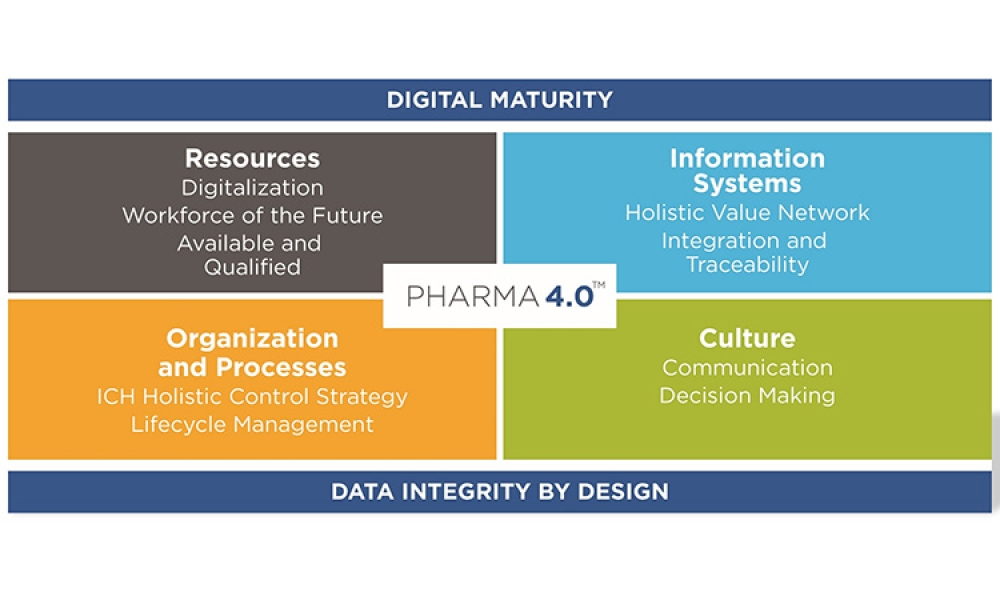
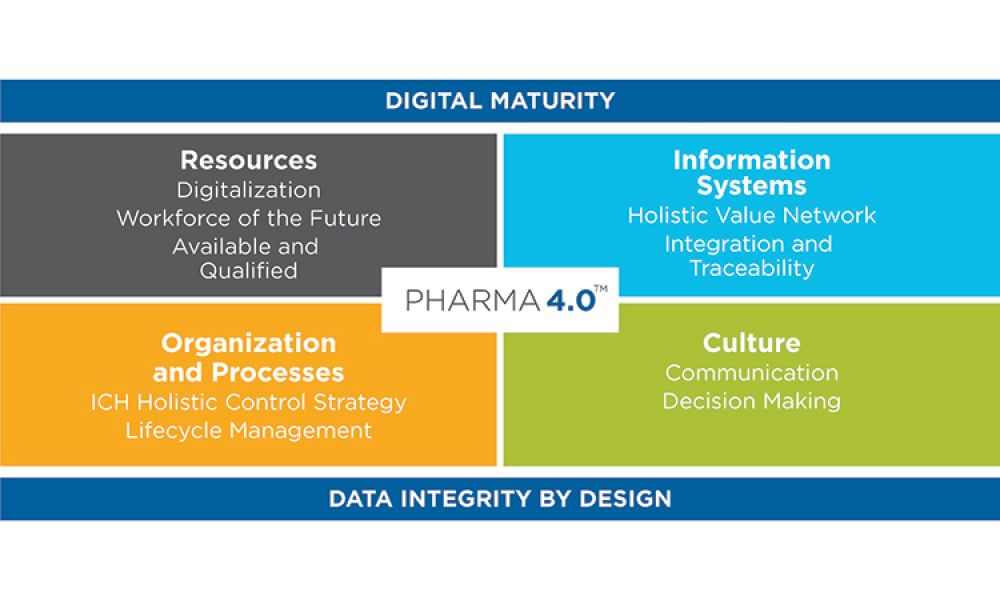
This article summarizes the key findings from the 7th Pharma 4.0™ Survey, conducted in 2023. It explores the demographics of the survey respondents, maturity levels of Pharma 4.0™ adoption, enabling technologies being leveraged, anticipated benefits, and challenges encountered.